CASE PRESENTATION
A 52-year-old man with no previous ophthalmic history presented to the New York Eye and Ear Infirmary of Mount Sinai Glaucoma Service with an IOP of 50 mm Hg in his right eye. The patient stated that he had experienced blurry vision and intermittent pain in his right eye for the past 3 months. He denied any antecedent trauma or surgery. His past medical history was significant only for hypertension and coronary artery disease, for which he took amlodipine, metoprolol, valsartan, and simvastatin.
Upon examination, the patient's visual acuity with correction (-2.00 D sphere OU) was 20/40 OD and 20/20 OS. No relative afferent pupillary defect was noted. A slit-lamp examination of the anterior segment of the right eye revealed 3+ pigmented cells in the anterior chamber and a diffuse scattering of pigment on the corneal endothelium. There were no retroillumination defects noted or any other abnormalities of the iris. An examination of the left eye was within normal limits. Gonioscopy of the right eye demonstrated a Shaffer grade 4 open angle with a thick band of clumped pigment in all quadrants overlying both the scleral spur and ciliary body, whereas the left eye revealed a Shaffer grade 4 angle with normal pigmentation of the trabecular meshwork (Figure 1). On dilated fundoscopic examination, the cup-to-disc ratio was 0.5 OD and 0.3 OS. The results of the peripheral retinal examination were within normal limits. Ultrasound biomicroscopy (UBM) demonstrated a flat iris contour with no contact between the posterior aspect of the iris and zonular fibers in either eye. No masses of the iris or ciliary body were identified.
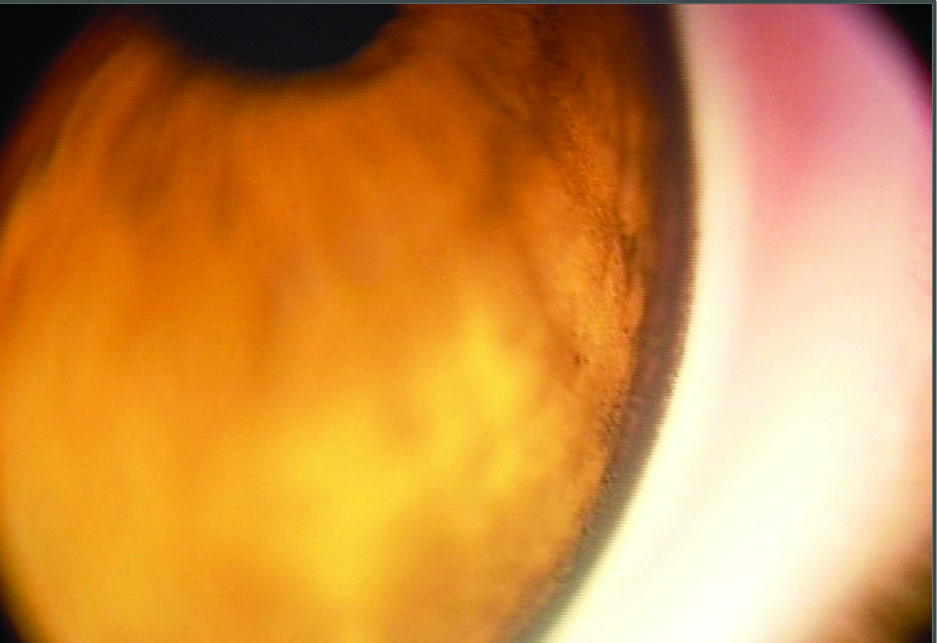
Figure 1. (A) Gonioscopic view of the temporal angle of the right eye. The angle is wide open, and the trabecular meshwork and adjacent structures are heavily pigmented.

Figure 1. (B) A gonioscopic view of the temporal angle of the left eye. Note that the angle is open to the ciliary body band, and the trabecular meshwork is lightly pigmented
HOW WOULD YOU PROCEED?
- Would you treat this as pigment dispersion syndrome?
- Would you perform further testing? If so, for what would you test?
- Would you refer the patient to another subspecialist?
CLINICAL COURSE
Given that this was a unilateral case of pigment dispersion with no antecedent trauma, the main concern was to evaluate the patient for the presence of a melanocytic tumor. Although UBM did not initially reveal a mass, we referred the patient to an ocular oncologist, as there was a high suspicion of malignancy. The patient was started on timolol-brimonidine (Combigan; Allergan) twice daily and latanoprost at night in his right eye as well as 500 mg of oral acetazolamide twice daily.
Two weeks later, at his visit to the ocular oncologist, B-scan ultrasonography revealed the presence of a ciliary body mass at the 12-o'clock position. UBM was repeated, and the mass was noted to have a width of 6.89 mm, a height of 5.04 mm, and a depth of 4.39 mm (Figure 2).
The differential diagnosis included melanocytoma, nevus, melanoma, adenoma, and medulloblastoma. The patient was offered two options: excisional biopsy or observation with re-evaluation in 3 to 4 weeks. When the patient returned 3 weeks later, the size of the mass had increased to 9 mm by 4.5 mm, leading us to conclude that this was likely a ciliary body melanoma (Figure 3). Two weeks later, the patient underwent plaque brachytherapy using ruthenium 106 (Figure 4). A total of 86 Gy were applied over 4 days. On the fifth day, the plaque was surgically removed.
At the most recent follow-up visit, the patient's condition remained stable, and only topical therapy was required to control the IOP.
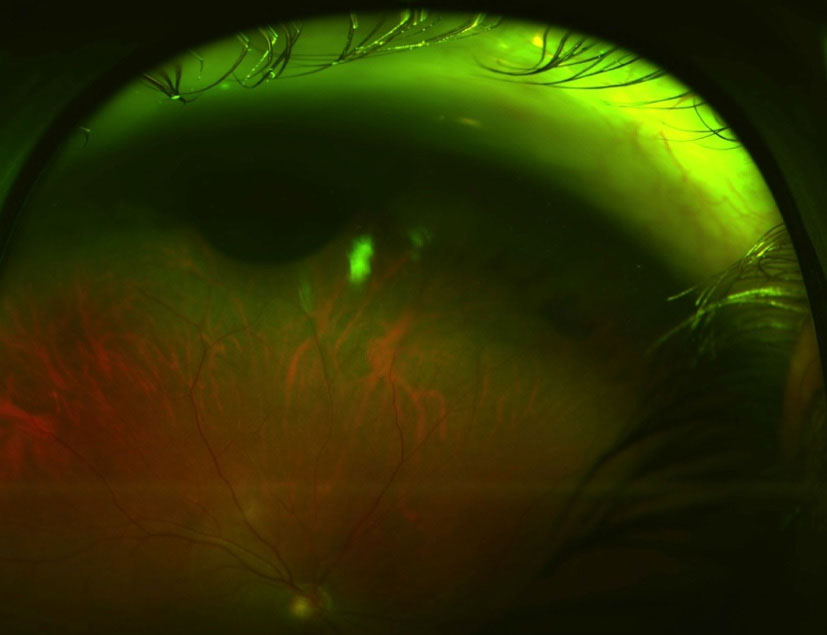
Figure 3. A melanocytic lesion is positioned anteriorly at 12 o'clock.
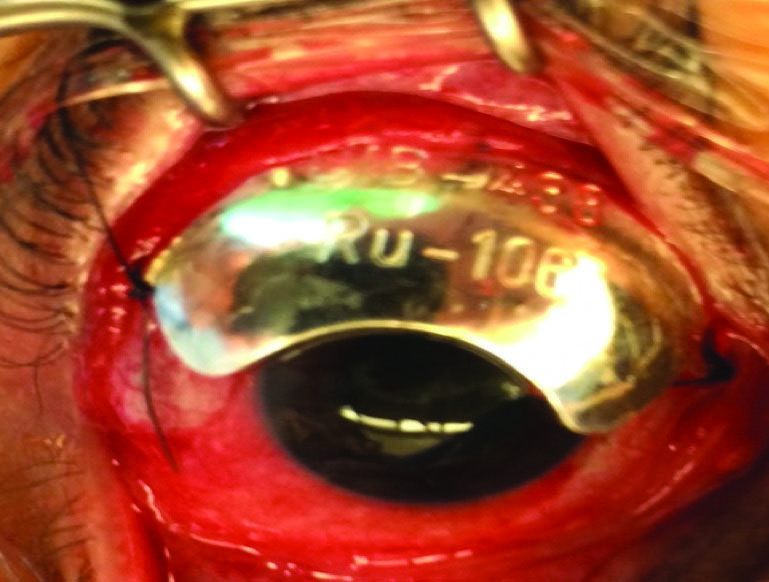
Figure 4. Ruthenium plaque placement.
DISCUSSION
Patients with pigmented tumors of the uveal tract such as melanomas and melanocytomas can present with unilateral findings that can mimic pigment dispersion syndrome. On examination, several distinguishing findings allow the clinician to make the correct diagnosis. The presence of pigment on the corneal endothelium, pigment in the anterior chamber, and a heavily pigmented trabecular meshwork are common, nonspecific findings that many conditions share. Laterality, iris appearance, and iris configuration need to be evaluated carefully to help differentiate true pigment dispersion syndrome from a masquerade syndrome that can be life-threatening.
Pigmentary glaucoma can result from the presence of a ciliary body or choroidal melanoma. When these tumors come in contact with the iris, pigment is liberated and will circulate through the anterior chamber and collect in the trabecular meshwork. This can reduce aqueous outflow and lead to sudden, temporary elevations in IOP. As the pigment deposits over a longer period of time, there can be pathologic changes to the trabecular endothelial cells, which lead to a chronic, secondary glaucoma. Pigmentladen macrophages can also obstruct the trabecular meshwork, and when this is the case, the condition is more properly referred to as melanomalytic glaucoma. Clinical examination typically reveals a significant amount of pigment granules in the anterior chamber and a scattered dusting of pigment on the posterior surface of the cornea. On gonioscopy, there is irregular clumping rather than a homogenous distribution of pigment in the trabecular meshwork. The iris will have a normal contour (although focal anterior bulging with peripheral synechiae formation may be present due to mass effect from the tumor), and no retroillumination defects are typically found. In contrast, patients with pigment dispersion syndrome will have a midperipheral iris concavity that can be best seen on gonioscopy and UBM. The posteriorly bowed iris rubs directly against the zonular fibers, resulting in pigment release. This pigment disruption is what accounts for the midperipheral iris retroillumination defects that are characteristic of the condition. However, these findings are not present in every case. Although clinical examination findings are especially helpful in differentiating between the two conditions, laterality may be of the greatest significance. Pigmentary glaucoma that results from the presence of an intraocular melanocytic tumor will present with unilateral findings, whereas clinical signs of pigment dispersion syndrome will manifest in both eyes, although the glaucomatous damage may be asymmetric.
Once the diagnosis of melanoma is made, treatment should be initiated immediately and typically involves radiation and/or surgery. Radiation can be in the form of either brachytherapy or external beam particle therapy. Complications can include ischemic retinopathy, optic neuropathy, cataract, neovascular glaucoma, and vitreous hemorrhage.1,2 Surgery can involve either local resection or enucleation. Other less commonly used therapies include photocoagulation, transpupillary thermotherapy, and photodynamic therapy. If metastasis has occurred, systemic treatment is needed.3-8
The management of glaucoma in patients with an intraocular tumor is challenging. Incisional surgery is typically contraindicated, given the risk of extraocular tumoral spread. Pharmacologic therapy is instituted first and maximized until the IOP is controlled. If the IOP remains elevated, laser trabeculoplasty or transscleral cyclophotocoagulation can be attempted. Ultimately, much depends upon the patient's long-term prognosis. If pain is an issue due to uncontrolled IOP and the eye has poor potential, retrobulbar alcohol or enucleation may be considered.
CONCLUSION
The case described herein serves as a reminder that many disease processes can present as secondary glaucoma. It is especially important for eye care specialists to recognize those conditions that are not only sight threatening but also life-threatening to patients. In this case, a uveal melanoma masqueraded as pigment dispersion syndrome. The unilateral nature of the presentation with an elevated IOP and pigment clumping in the anterior chamber angle without a prior history of trauma strongly pointed to the possibility of a melanocytic mass. This finding prompted further investigation with UBM and referral to an ophthalmic oncologist, who helped confirm the diagnosis and institute treatment.
Yijie (Brittany) Lin, MD, is a resident at the New York Eye and Ear Infirmary of Mount Sinai in New York. Dr. Lin acknowledged no financial interest in the products or companies mentioned herein.
Joseph F. Panarelli, MD, is an ophthalmologist at the New York Eye and Ear Infirmary of Mount Sinai in New York. He acknowledged no financial interest in the products or companies mentioned herein. Dr. Panarelli may be reached at jpanarelli@nyee.edu.
- Gragoudas ES, Li W, Lane AM, et al. Risk factors for radiation maculopathy and papillopathy after intraocular irradiation. Ophthalmology. 1999;106(8):1571-1578.
- Kellner U, Bornfeld N, Foerster MH. Radiation-induced optic neuropathy following brachytherapy of uveal melanomas. Graefes Arch Clin Exp Ophthalmol. 1993;231(5):267-270.
- Karcioglu ZA, Haik BG. Eye, orbit, and adnexal structures. In: Abeloff MD, Armitage JO, Niederhuber JE, et al, eds. Abeloff's Clinical Oncology. Philadelphia, PA; Churchill, Livingstone, Elsevier: 2008.
- Gragoudas E, Li W, Goiten M, et al. Evidence-based estimates of outcomes in patients irradiated for intraocular melanoma. Arch Ophthalmol. 2002;120(12):1665-1671.
- Journee-de Korver JG, Oosterhuis JA, de Wolff-Rouendaal D, Kemme H. Histopathological findings in human choroidal melanomas after transpupillary thermotherapy. Br J Ophthalmol. 1997;81:234-239.
- vanBeek JGM, Koopmans AE, Verdiik RM, et al. Diagnosis, histopathologic and genetic classification of uveal melanoma. In: Huynh Thien Duc G, ed. Melanoma: From Early Detection to Treatment. Rijeka, Croatia; Intech: 2013.http://www.intechopen.com/books/melanoma-from-early-detection-to-treatment/diagnosis-histopathologicand- genetic-classification-of-uveal-melanoma.
- Shields CL, Shields JA, Cater J, et al. Plaque radiotherapy for uveal melanoma: long-term visual outcome in 1,106 consecutive patients. Arch Ophthalmol. 2000;118(9):1219-1228.
- Johnson DL, Altaweel MM, Neekhra A, et al. Uveal melanoma masquerading as pigment dispersion glaucoma. Arch Ophthalmol. 2008;126(6):866-876.
