Current treatments for the elevated IOP that is a hallmark of most forms of glaucoma focus either on lowering the rate of aqueous humor inflow or increasing the flow of aqueous humor through the secondary, unconventional outflow pathway. Because the pathology responsible for ocular hypertension (OHT) is located in the primary, conventional outflow pathway, it would be preferable to identify treatments that target this diseased tissue, but where in the aqueous outflow system is the locus of increased resistance to flow in glaucoma?
The primary resistance to the flow of aqueous humor through the conventional outflow pathway is thought to be generated in the trabecular meshwork—specifically in the extracellular matrix filling the open space of the juxtacanalicular connective tissue (JCT). After the aqueous humor traverses the JCT, it passes through pores in the endothelial lining of the inner wall of Schlemm canal (SC) that abuts the JCT. Although flow resistance through these pores is negligible, they have a hydrodynamic interaction with the JCT—called the funneling effect—in which pore density modulates the effective flow resistance of the JCT.1,2
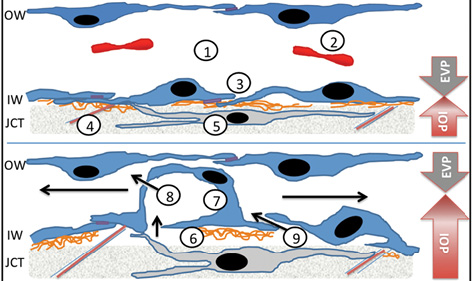
Figure 1. Dynamic biomechanical responses of SC endothelia to a pressure gradient: 1–SC lumen; 2–red blood cell; 3–intercellular tight junction; 4–elastin fiber; 5–JCT-trabecular meshwork cell; 6–discontinuous basement membrane; 7–cellular outpouching known as a “giant vacuole” in an SC cell; 8–flow through intracellular pore; 9–flow through paracellular pore. Black arrows indicate the direction of flow. Abbreviations: OW, outer wall; IW, inner wall; EVP, episcleral venous pressure.
Because of this hydrodynamic interaction, either a decreased permeability of the extracellular matrix in the JCT or a reduced density of pores in the inner-wall endothelium could cause the increased flow resistance characteristic of glaucoma. An optimal strategy for treating OHT therefore needs to be directed at one or both of these possibilities. Although accumulations of abnormal components of extracellular matrix have been reported in glaucoma, they do not appear to account for the increased resistance to outflow.3,4 Decreased pore density, however, has been described in glaucomatous eyes.5,6 Moreover, several recent findings regarding the mechanobiology of SC cells have now elucidated the mechanism by which altered cytoskeletal mechanics can lead to decreased pore formation, OHT, and glaucoma.
unique mechanical environment of SC cells
The inner wall of SC has two critical responsibilities in a mechanically demanding environment that, on the surface, appear to be in opposition. SC cells of the inner wall need to maintain ocular immune privilege by preserving the blood-aqueous barrier. At the same time, they help regulate resistance to fluid flow out of the eye, thus sustaining a reliable IOP. Several unique features have evolved to accomplish these tasks.
Distinctively, fluid flow across the inner wall of SC is in the basal-to-apical direction (Figure 1), the opposite of other blood microvascular endothelial beds but similar to terminal lymphatics that make relatively leaky junctions.2 Uniquely, SC is a blood-lymphatic hybrid, expressing selective protein markers of each.7,8 Such an arrangement facilitates the “one-way–valve” behavior of SC and pressure-driven flow of aqueous humor into the systemic venous circulation, while preventing blood products from accessing the interior of the eye during daily elevations of episcleral venous pressure.
To properly function in this challenging mechanical environment (Figure 1), SC
• is impressively contractile9
• is anchored to underlying tissues via a discontinuous basement membrane and attachments to an elastin network
• dynamically forms balloon-like “outpouchings” into the canal lumen
• develops micron-sized paracellular and transcellular openings that serve as “release valves” (called pores) to prevent cell detachment
All of these unique adaptations provide opportunities for selective therapeutic targeting in people with glaucoma.
IMPORTANCE OF CYTOSKELETAL MECHANICS TO AQUEOUS OUTFLOW
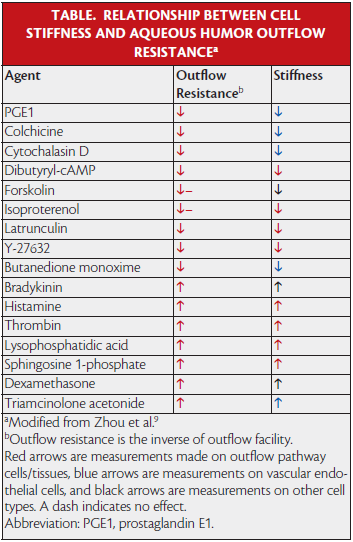
Zhou et al examined the effect of a number of agents known to affect outflow resistance on the biomechanical behavior of SC cells.9 Remarkably, agents that decrease outflow resistance (and thereby IOP) uniformly decrease the stiffness of SC cells, whereas drugs that increase outflow resistance do the contrary (Table). SC cells also possess an extraordinary dynamic contractile range, which appears adaptive for functioning in their challenging biomechanical environment and is comparable to that of smooth muscle cells in the lung (Figure 2).
Recently, Overby et al studied how the biomechanical behavior of SC cells might be altered in glaucomatous cell strains.10 The investigators found that glaucomatous SC cells are stiffer than normal and that this characteristic negatively correlated with their pore-forming capability (Figure 3). These data strongly support the hypothesis that altered cell stiffness in glaucoma decreases pore formation and increases outflow resistance.
Overby et al considered what might cause the greater stiffness of SC cells and found that glaucomatous SC cells are particularly sensitive to substrate stiffness. In other words, glaucomatous SC cells more dramatically increase their stiffness when placed on stiff substrates as compared with a smaller effect in healthy SC cells. These findings are particularly relevant, because in glaucomatous eyes, it appears that the underlying trabecular meshwork is much stiffer than normal.11 Furthermore, Overby et al found that, when grown on substrates simulating the stiffer trabecular meshwork found in glaucoma, SC cells from glaucomatous eyes greatly increase their expression of connective tissue growth factor, a protein already associated with OHT and outflow obstruction (Figure 4).12
These findings have significant implications with respect to drugs currently in clinical trials for IOP reduction such as Rho kinase inhibitors,13 actin depolymerizers,14 adenosine A1 agonists,15 and nitric oxide donors16 that all act to relax SC cells and consequently improve conventional outflow. Importantly, these drugs work additively with existing drugs and are expected to restore function to the diseased, pressure-dependent, conventional outflow cells/tissue responsible for OHT in glaucoma. Clinical outcomes for glaucoma patients are therefore expected to be measurably improved compared with currently available medical therapy.
Although promising, three of the four classes of drugs currently in clinical trials interact with proteins that are widely expressed, so the agents carry the potential for undesirable side effects. It will be interesting to see the results from clinical trials.13,14 New classes of ocular hypotensive drugs that specifically target SC cells are therefore needed, because they offer the promise of fewer off-target effects. Although the precise mechanism of action is unknown, recent studies have demonstrated that drugs can be developed that target SC and not trabecular meshwork cells.17 Identifying compounds that interact specifically with SC cells and decrease outflow resistance will also provide important insight into the cellular control of outflow resistance and enhance understanding of its dysfunction in glaucoma. The targeted delivery of agents that relax SC cells can thus be expected to optimize the treatment of the diseased tissue in glaucoma.
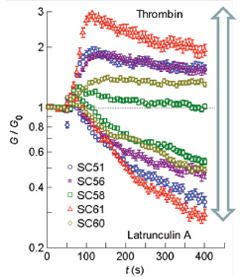
Figure 2. Changes in the contractile state of SC cells (G/G0) after application of a relaxing agent (latrunculin) or contractile agonist (thrombin). The contractile range was quite large.9
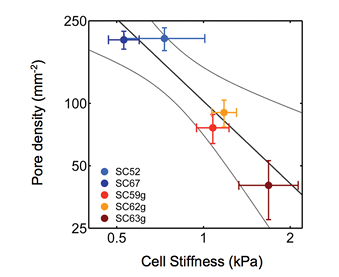
Figure 3. Correlation (dark line) between pore density and cell stiffness, as measured by atomic force microscopy in two normal SC cell strains (SC52, SC67) and three glaucomatous SC cell strains (SC50g, SC62g, SC64g). Bars represent standard error of the mean of pore density and modulus. Light curves represent 95% confidence intervals.10
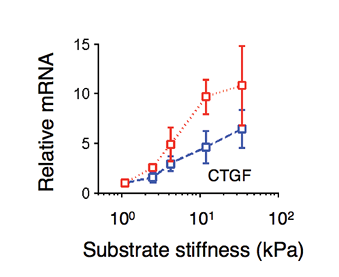
Figure 4. Increases in substrate stiffness differentially modulated connective tissue growth factor (CTGF) expression between normal SC cell strains (blue) and glaucomatous SC cell strains (red). All expression levels were normalized to that on the softest gel. Mean ±SEM.10
Exploiting SC cells’ unique features for SELECTIVE therapeutic targeting
Genetic and functional high-throughput screens are in development to facilitate the discovery of SC-selective drugs. These assays attempt to take advantage of unique features and behaviors of SC. High-throughput assays have been developed based on a technique called traction force microscopy that monitors the contractile state of a cell. These assays efficiently measure drug effects on SC’s dynamic range of contractility.18 Fluorescence-based assays were designed to detect drugs that increase pore formation in SC monolayers.19 Hydraulic conductivity assays using novel patterned scaffolds monitor drug effects on basal-to-apical fluid flow across SC monolayers.20
In addition, recent studies have demonstrated quantifiable differences between SC cells isolated from healthy and glaucomatous donor eyes,10 variations that likely have a genetic or epigenetic basis. Expression profiling and characterization of DNA open reading frames and methylation patterns from these cells is therefore underway, and they hold promise for the identification of signaling network “nodes” or expression changes that may underlie aberrant behavior. Such vulnerabilities in glaucomatous SC cells represent opportunities for drug targeting or gene therapy to restore the function of diseased outflow tissue in patients. n
Mark Johnson, PhD, is a professor in the Departments of Biomedical Engineering, Mechanical Engineering, and Ophthalmology at Northwestern University in Evanston, Illinois. Dr. Johnson may be reached at m-johnson2@northwestern.edu.
W. Daniel Stamer, PhD, is a professor of ophthalmology and biomedical engineering at Duke University in Durham, North Carolina. Dr. Stamer may be reached at dan.stamer@duke.edu.
1. Johnson M, Shapiro A, Ethier CR, Kamm RD. Modulation of outflow resistance by the pores of the inner wall endothelium. Invest Ophthalmol Vis Sci. 1992;33:1670-1675.
2. Overby D, Stamer D, Johnson M. The changing paradigm of outflow resistance generation: towards synergistics models of the JCT and inner wall endothelium. Exp Eye Res. 2009;88:656-670.
3. Ethier CR, Kamm RD, Palaszewski BA, et al. Calculations of flow resistance in the juxtacanalicular meshwork. Invest Ophthalmol Vis Sci. 1986;27:1741-1750.
4. Murphy CG, Johnson M, Alvarado JA. Juxtacanalicular tissue in pigmentary and primary open angle glaucoma. The hydrodynamic role of pigment and other constituents. Arch Ophthamol. 1992;110:1779-1785.
5. Allingham RR, Dekater AW, Ethier CR, et al. The relationship between pore density and outflow facility in human eyes. Invest Ophthalmol Vis Sci. 1992;33:1661-1669.
6. Johnson M, Chan D, Read AT, et al. The pore density in the inner wall endothelium of Schlemm’s canal of glaucomatous eyes. Invest Ophthalmol Vis Sci. 2002;43:2950-2955.
7. Ramos R, Hoying J, Witte M, Stamer D. Schlemm’s canal endothelia, lymphatic, or blood vasculature. J Glaucoma. 2007;16:391-405.
8. Kizhatil K, Ryan M, Marchant JK, et al. Schlemm’s canal is a unique vessel with a combination of blood vascular and lymphatic phenotypes that forms by a novel developmental process. PLoS Biol. 2014;12:e1001912.
9. Zhou EH, Krishnan R, Stamer WD, et al. Mechanical responsiveness of the endothelial cell of Schlemm’s canal: scope, variability and its potential role in controlling aqueous humour outflow. J R So Interface. 2012;9:1144-1155.
10. Overby DR, Zhou EH, Vargas-Pinto R, et al. Altered mechanobiology of Schlemm’s canal endothelial cells in glaucoma. Proc Natl Acad Sci USA. 2014;111:13876-13881.
11. Last JA, Pan T, Ding Y, et al. Elastic modulus determination of normal and glaucomatous human trabecular meshwork. Invest Ophthalmol Vis Sci. 2011;52:2147-2152.
12. Junglas B, Kuespert S, Seleem AA, et al. Connective tissue growth factor causes glaucoma by modifying the actin cytoskeleton of the trabecular meshwork. Am J Pathol. 2012;180:2386-2403.
13. Williams RD, Novack GD, van Haarlem T, Kopczynski C, Group ARPAS. Ocular hypotensive effect of the Rho kinase inhibitor AR-12286 in patients with glaucoma and ocular hypertension. Am J Ophthalmol. 2011;152:834-841.
14. Chen J, Runyan SA, Robinson MR. Novel ocular antihypertensive compounds in clinical trials. Clin Ophthalmol. 2011;5:667-677.
15. A phase II study to evaluate the additivity of trabodenoson to lataonoprost in adults with ocular hypertension or primary open-angle glaucoma. Clinical trials identifier: NCT01917383. Updated July 29, 2014. https://clinicaltrials.gov/ct2/show/NCT01917383. Accessed April 15, 2015.
16. Krauss AH, Impagnatiello F, Toris CB, et al. Ocular hypotensive activity of BOL-303259-X, a nitric oxide donating prostaglandin F2alpha agonist, in preclinical models. Exp Eye Res. 2011;93:250-255.
17. Wang JW, Woodward DF, Stamer WD. Differential effects of prostaglandin E2-sensitive receptors on contractility of human ocular cells that regulate conventional outflow. Invest Ophthalmol Vis Sci. 2013;54:4782-4790.
18. Park C-Y, Zhou EH, Tambe D, et al. High-throughput screening for modulators of cellular contractile force. Integrative Biology. In press.
19. Braakman S, Stamer W, Overby D. A fluorescent permeability assay for Schlemm’s canal endothelial cells in response to stretch. Presented at: Association for Research in Vision and Ophthalmology Annual Meeting; 2014; Orlando, FL; e-abstract 5983.
20. Dautriche C, Torrejon K, Bergkvist M, Xie Y, Sharfstein S, Danias J. 3D-Bio-engineering of the conventional outflow tract for high throughput glaucoma drug screening. Presented at: Association for Research in Vision and Ophthalmology Annual Meeting; 2014; Orlando, FL; e-abstract 5659.
