Optical coherence tomography (OCT) has become an indispensible tool in the armamentarium of eye care practitioners for the diagnosis and monitoring of glaucomatous optic neuropathy. Earlier time-domain OCT platforms allowed clinicians to reliably distinguish glaucomatous from nonglaucomatous eyes.1 Limited reproducibility precluded the use of this technology for detecting glaucomatous progression. The advent of spectral-domain OCT (SD-OCT) markedly improved scan reproducibility and the ability to detect glaucomatous progression on the basis of retinal nerve fiber layer (RNFL) thinning and optic nerve head (ONH) deterioration over time.2
MACULAR THICKNESS PROTOCOLS
Novel OCT-derived macular thickness protocols are now used to complement RNFL and ONH measurements in the diagnosis of glaucoma.3 These macular protocols are commercially available as the Ganglion Cell Complex Analysis (RTVue FD-OCT; Optovue), the Posterior Pole Asymmetry Analysis (Spectralis SD-OCT System; Heidelberg Engineering), and the Ganglion Cell Analysis (Cirrus HD-OCT; Carl Zeiss Meditec).
Current evidence supports the potential role of SD-OCT–derived macular thickness protocols in the assessment of glaucomatous progression. In a study investigating variation in the thickness of the ganglion cell-inner plexiform layer (GC-IPL) over time in clinically stable glaucoma patients, Kim and colleagues found that excellent intervisit reproducibility could be achieved.4 Reproducibility was highest and lowest for the average and minimum GC-IPL thickness measurements, respectively. Naghizadeh et al recently performed a study investigating the use of various RNFL, ONH, and ganglion cell complex (GCC) parameters to detect structural change in patients with early glaucomatous progression diagnosed on the basis of visual field change.5 The researchers found that the pattern-based GCC parameters (GCC focal loss volume and GCC global loss volume) were better able to detect progressive change when compared with RNFL, ONH, and average GCC measurements.
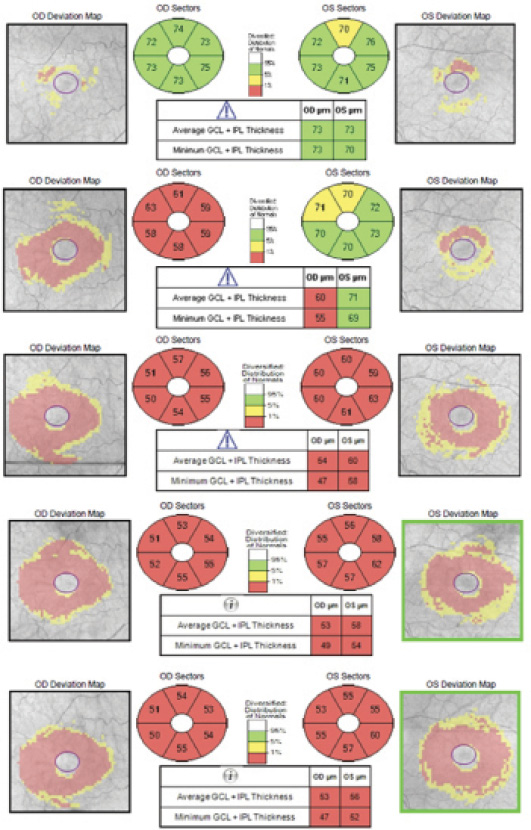
Figure. Serial SD-OCT scans of the macula demonstrating progressive thinning of the GC-IPL of each eye.
MONITORING ADVANCED DISEASE
An especially important role for SD-OCT–derived macular thickness protocols may be in the determination of progression in cases of advanced glaucoma. In these cases, determination of progression on the basis of automated visual field testing is often limited by fluctuations that are likely unrelated to the underlying disease.6 Furthermore, OCT-derived peripapillary RNFL measurements are often no longer useful in cases of advanced disease, because significant loss of this layer leaves behind a residual “floor” that no longer changes with ongoing glaucomatous progression.7 On the other hand, postmortem studies suggest that viable cells remain in the retinal ganglion cell layer in patients who have suffered advanced glaucomatous damage.8
In a study of 98 eyes of 98 patients with advanced glaucoma, Sung and colleagues found that peripapillary RNFL measurements obtained with SD-OCT did not differ between patient groups deemed to have stable versus progressive disease as determined by three expert glaucoma specialists.9 Macular thickness measured with SD-OCT, however, showed significantly higher rates of loss in patients with progressive versus stable disease. The investigators postulated that the papillomacular bundle is more resistant to glaucomatous damage than the peripapillary RNFL and therefore may continue to show change in late stages of the disease.
LIMITATIONS
It is imperative for clinicians to be aware of the potential limitations of using OCT-measured macular thickness to detect glaucomatous progression. Most importantly, macular thickness is known to demonstrate continual thinning with increasing age in nonglaucomatous individuals.10 Clinicians must take into account the degree of expected physiologic macular thinning when using macular thickness change to determine the presence or absence of glaucomatous progression. In a study of 150 eyes of 90 patients with glaucoma and 72 eyes of 40 nonglaucomatous individuals, Leung et al estimated the mean rate of change of GC-IPL thickness to be -0.318 µm/year.10 The group observed a notable reduction in the proportion of eyes with progressive disease on the basis of macular parameters after accounting for this age-related change. The change in the proportion of progressing eyes was much less when using peripapillary RNFL measurements. The investigators concluded that age-related changes affect macular thickness measurements much more than peripapillary RNFL measurements.
CONCLUSION
Progressive changes in macular thickness, as measured by SD-OCT, may indicate the presence of glaucomatous progression (Figure). This parameter may be most useful in cases of advanced glaucoma, when visual field tests and peripapillary RNFL measurements are often of limited utility. Physicians must take into account physiologic age-related changes when using this parameter in clinical decision making. n

Ahmad A. Aref, MD, is an assistant professor of ophthalmology at the Illinois Eye & Ear Infirmary, University of Illinois at Chicago School of Medicine, Chicago. He acknowledged no financial interest in the products or companies mentioned herein. Dr. Aref may be reached at (312) 996-7030; ahmadaref@gmail.com.
1. Hougaard JL, Heijl A, Bengtsson B. Glaucoma detection by Stratus OCT. J Glaucoma. 2007;16(3):302-306.
2. Leung CK. Diagnosing glaucoma progression with optical coherence tomography. Curr Opin Ophthalmol. 2014;25(2):104-111.
3. Tan O, Chopra V, Lu AT, et al. Detection of macular ganglion cell loss in glaucoma by Fourier-domain optical coherence tomography. Ophthalmology. 2009;116(12):2305-2314.
4. Kim KE, Yoo BW, Jeoung JW, Park KH. Long-term reproducibility of macular ganglion cell analysis in clinically stable glaucoma patients [published online ahead of print March 31, 2015]. Invest Ophthalmol Vis Sci. doi:10.1167/iovs.
5. Naghizadeh F, Garas A, Vargha P, Hollo G. Detection of early glaucomatous progression with different parameters of the RTVue optical coherence tomograph. J Glaucoma. 2014;23(4):195-198.
6. Advanced Glaucoma Intervention Study. 2. Visual field test scoring and reliability. Ophthalmology. 1994;101(8):1445-1455.
7. Mwanza JC, Budenz DL, Warren JL, et al. Retinal nerve fibre layer thickness floor and corresponding functional loss in glaucoma. Br J Ophthalmol. 2015;99(6):732-737.
8. Pavlidis M, Stupp T, Naskar R, et al. Retinal ganglion cells resistant to advanced glaucoma: a postmortem study of human retinas with the carbocyanine dye Dil. Invest Ophthalmol Vis Sci. 2003;44(12):5196-5205.
9. Sung KR, Sun JH, Na JH, et al. Progression detection capability of macular thickness in advanced glaucomatous eyes. Ophthalmology. 2012;119(2):308-313.
10. Leung CK, Ye C, Weinreb RN, et al. Impact of age-related change of retinal nerve fiber layer and macular thicknesses on evaluation of glaucoma progression. Ophthalmology. 2013;120(12):2485-2492.
