Patients with advanced glaucoma require more careful monitoring than those with mild or moderate disease. The former are more susceptible to further vision loss and generally require lower IOPs than those with early or mild vision loss. In addition, progressive vision loss is usually more noticeable and debilitating in patients with late- versus early-stage disease. Furthermore, the instruments that assess optic nerve damage and visual field loss may be less sensitive to changes in eyes with severe disease. For these reasons, monitoring patients with advanced glaucoma requires special considerations and adjustments to eye care specialists’ usual methods.
DEFINING ADVANCED GLAUCOMA
Despite a lack of consensus on the definition of advanced glaucoma, several sources provide some guidance. According to the classic Hodapp-Parrish-Anderson textbook, patients with a mean deviation of -12 dB on the standard Humphrey Visual Field 24-2 program (Carl Zeiss Meditec) have advanced glaucoma.1 The International Classification of Diseases 9 (365.73) and 10 (7th digit “3”) diagnostic code defines severe-stage, advanced-stage, and end-stage glaucoma as optic nerve abnormalities consistent with glaucoma and glaucomatous visual field abnormalities in both hemifields and/or loss within 5º of fixation in at least one hemifield. Regardless of the precise definition, patients with advanced glaucoma likely suffer from some visual disability that impairs their ability to perform activities of daily living.
FREQUENCY OF VISITS
Given the severe and tenuous nature of their vision loss, patients with advanced glaucoma require frequent visits to monitor their IOP control, their optic nerve status, and their visual functioning. I have seen patients lose significant vision in just a few months between visits when their long-standing IOP control suddenly falters. I therefore generally see my patients with advanced glaucoma every 3 to 4 months, even if I am satisfied with their IOP control.
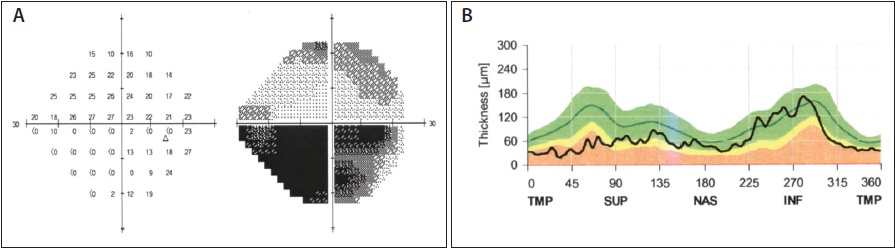
Figure. Advanced glaucoma in a patient’s right eye. The visual field shows almost complete loss of her inferior hemifield (A). The thickness of the corresponding spectral domain optical coherence tomography retinal nerve fiber layer (SD-OCT RNFL) has reached a floor at approximately 40 to 50 μm. Note that her inferior RNFL can still be monitored by SD-OCT (B).
VISUAL FIELDS
In the advanced stages of glaucoma, the standard 24-2 visual fields may no longer be very sensitive to subtle progression. This strategy will miss defects that develop or progress between the 6º spacing of the test points. The outer field area may already be gone, and too much time will be wasted waiting for the patient to see a stimulus. In many cases, a 10-2 strategy that tests the central 10º with 2º spacing is more sensitive to visual field changes. Alternatively, a larger stimulus size V can be used on the 24-2 program.
It is also important to remember that, for patients with advanced visual field loss, the visual fields may be more highly variable, particularly at the borders of existing defects. Repeat testing to confirm changes is recommended.
OPTIC NERVE ASSESSMENT
Detecting glaucomatous changes in the optic nerve is more challenging in eyes with an already thin neuroretinal rim and a 0.9 cup-to-disc ratio than in eyes with thick rims and a 0.6 cup-to-disc ratio. Even with high-quality serial stereographic optic disc photographs, subtle changes are easy to miss in eyes with 0.9 cup-to-disc ratios. Even so, the optic nerve should still be carefully examined for disc hemorrhages, which may signal progressive damage.2
Imaging technologies such as SD-OCT also encounter problems with advanced optic nerve damage. On the SD-OCT measurements of the RNFL, the so-called floor effect becomes relevant in eyes with severe thinning. RNFL thinning levels off at approximately 40 to 50 µm, perhaps due to residual glial tissue, blood vessels, or other nonneural tissue.3 That said, remaining areas that are less damaged may still be monitored with SD-OCT (Figure). I generally rely more on visual fields than optic nerve imaging for observing patients with advanced glaucoma.
Weigh In on this Topic Now!
For the visual field testing of patients with advanced glaucoma, which testing strategy do you generally use?
A. A standard 24-2 testing strategy
B. A 24-2 testing strategy with an enlarged stimulus
C. A 10-2 testing strategy
D. None of the above
Answer here: https://www.surveymonkey.com/r/S7G369Y.
SUBJECTIVE ASSESSMENT
Among patients with advanced glaucoma, their own subjective assessment of their visual functioning is often the most important indicator of disease progression. Glaucoma’s reputation as the sneak thief of sight ceases to hold true in this population. Patients may notice subtle changes in their vision, and they are particularly sensitive to near-fixation defects. They may report that their vision is getting dimmer or that they need more light to read or perform daily activities. Additionally, they may report increased difficulty with reading or finding the next line of text on a page. In patients with vision loss from end-stage glaucoma, a drop in measured visual acuity may also be a sign of disease progression.
CONCLUSION
Certainly, it is preferable to detect glaucoma early and prevent its progression to the advanced stage. The reality of this disorder, however, is that a significant percentage of patients will present with advanced disease or will experience progression to advanced disease despite the best efforts of their eye care provider. For patients with advanced glaucoma, the visits are more frequent, the testing is more taxing, the treatment is more intensive, and most importantly, the risk of blindness is much higher. Monitoring patients with advanced glaucoma therefore requires an extra level of diligence on the clinician’s part. n

Darrell WuDunn, MD, PhD, is a professor of ophthalmology at the Eugene and Marilyn Glick Eye Institute, Indiana University School of Medicine, Indianapolis, Indiana. He acknowledged no financial interest in the product or company mentioned herein. Dr. WuDunn may be reached at (317) 274-2020; dwudunn@iupui.edu.
1. Hodapp E, Parrish RK II, Anderson DR. Clinical Decisions in Glaucoma. St. Louis, MO: Mosby; 1993.
2. Suh MH, Park KH. Pathogenesis and clinical implications of optic disk hemorrhage in glaucoma. Surv Ophthalmol. 2014;59(1):19-29.
3. Hood D, Kardon R. A framework for comparing structural and functional measurements of glaucomatous damage. Prog Retin Eye Res. 2007;26(6):688-710.
