
Ocular surface disease (OSD) is the biggest reason that people visit eye care practitioners.1-6 Learning more about how to detect, recognize, and effectively treat OSD can therefore benefit all ocular surgeons, because the condition is one of the leading causes of patients’ unhappiness or dissatisfaction after ocular surgery.
Glaucoma patients are at increased risk of developing OSD, because both diseases are more common in the elderly. In addition, the first line of glaucoma treatment in the United States is IOP-lowering eye drops, which often contain preservatives commonly associated with the development or worsening of OSD.7-9 The long-term use of topical medication containing preservatives such as benzalkonium chloride may damage the conjunctiva, cornea, and epithelial cells and decrease goblet cell density.10 Moreover, OSD is often associated with other ocular diseases such as blepharitis and meibomian gland dysfunction (MGD).
Recent advances in refractive and cataract surgery have revolutionized corneal imaging. Unfortunately, OSD can dramatically affect the accuracy of many of these measurements, which can adversely affect visual outcomes. This article reviews the principles and clinical significance of various corneal imaging technologies.
CORNEAL TOPOGRAPHY
Corneal topography used in clinical practice is most commonly based on Placido disc analysis. This method of imaging the anterior corneal surface relies on the analysis of reflected images of multiple concentric rings projected onto the cornea. Corneal topography is used most often to evaluate astigmatism and detect corneal pathology such as keratoconus and pellucid marginal degeneration.
Scanning-slit elevation evaluation is one elevation-based method of assessing topography. Via a combination of reflective corneal topography and optical slit design, the Orbscan (Bausch + Lomb) analyzes corneal thickness in addition to the anterior and posterior surfaces of the cornea.
Cassini (i-Optics) is a novel topographer employing multispot, multicolor light-emitting diode tear film-reflection imaging. Understanding corneal astigmatism helps to customize surgical planning.
CORNEAL TOMOGRAPHY
In corneal tomography, Scheimpflug-based devices generate images of the anterior eye with a camera positioned perpendicular to a slit beam so as to create an optical section of the cornea and lens. By including topography and pachymetry of the entire anterior and posterior surface of the cornea from limbus to limbus, the technology is able to detect early corneal irregularities such as ectasia and keratoconus. The four most relevant tomographic maps are the anterior sagittal curvature, anterior and posterior corneal elevation, and pachymetry maps. Corneal tomography can also accurately estimate anterior chamber dimensions, including angle width and anterior chamber depth.
ANTERIOR SEGMENT OPTICAL COHERENCE TOMOGRAPHY
Figure 1. The ArcScan Insight 100.
Anterior segment optical coherence tomography uses optical interferometry to produce a high-resolution, cross-sectional image of the cornea and other anterior segment structures. This noncontact technology requires no coupling medium, so operators can visualize the angle, iris, and lens in their natural state. The use of light as an analytical medium, however, may not allow adequate visualization if there are corneal opacities (eg, scarring or pannus) or deeper structures (eg, ciliary body), because the iris pigment epithelium absorbs the coherent light.
ULTRASOUND
High-frequency, high-resolution ultrasound biomicroscopy is a precise, noninvasive tool for imaging the anterior segment for diagnostic purposes and surgical planning. One application is for patients with narrow anterior chamber angles. Sound waves are not obstructed and can be used to view not only the angle but also structures posterior to the iris, including the ciliary sulcus, the ciliary body, the peripheral lens, the zonules, the IOL’s haptics, and the posterior chamber.11-13
The ArcScan Insight 100 (ArcScan; Figure 1) allows unprecedented accuracy in imaging and biometry of the eye.14-16 Indicated for refractive surgical planning and the evaluation of anterior segment pathology, this ultrasound device takes 100 images and several measurements, including anterior chamber depth, angle-to-angle width, individual corneal layers, and sulcus-to-sulcus width.
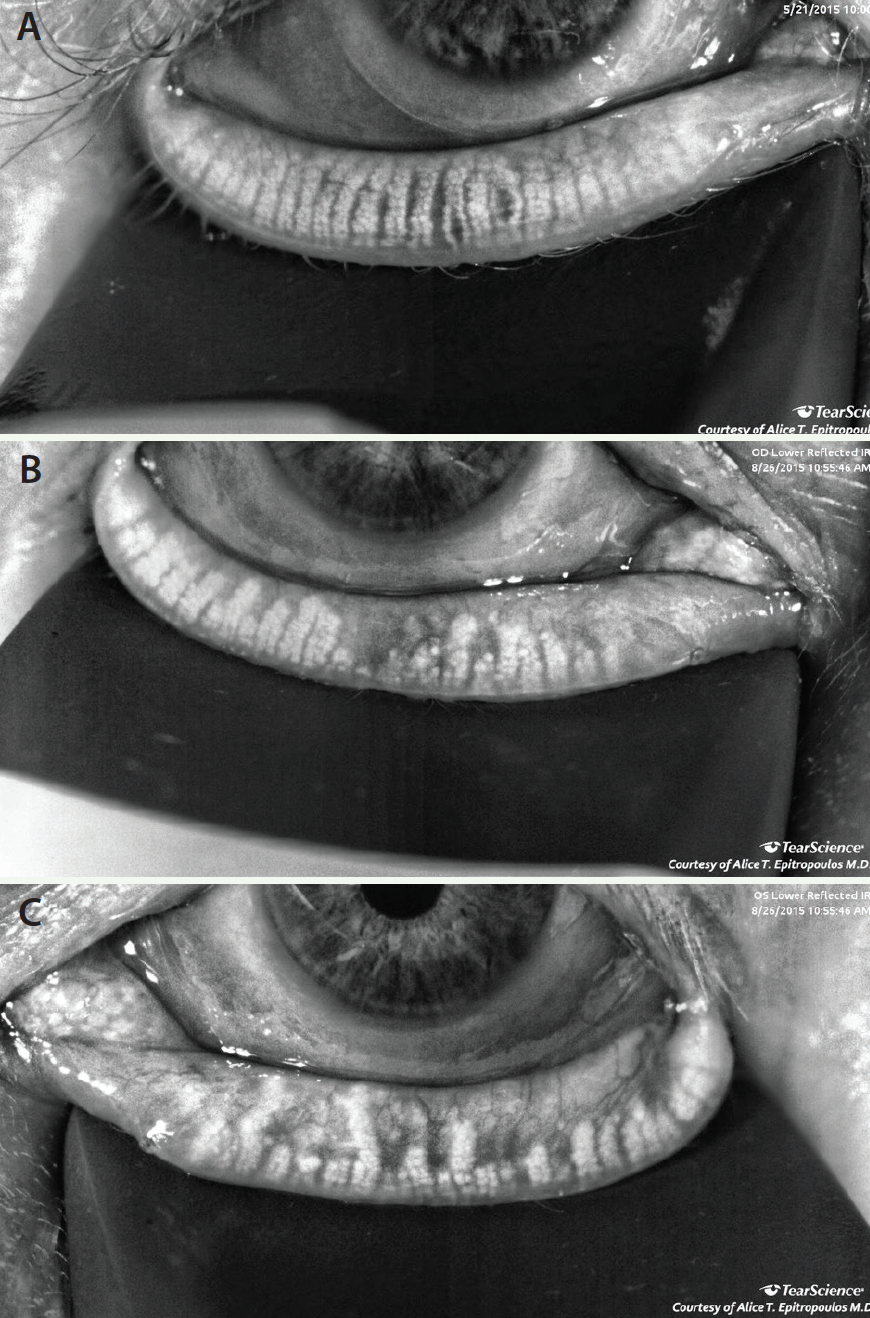
Figure 2. MGD with mild (A), moderate (B), and severe (C) atrophy.
MEIBOGRAPHY
Meibography provides high-definition images of the meibomian glands. MGD results in the stasis of meibum that, if left untreated, can lead to dilation and atrophy of the meibomian glands and a subsequent loss of function. Meibography is an excellent tool for identifying patients with MGD early as well as for educating them about the disease (Figure 2).
CONCLUSION
A thorough history and careful clinical examination are essential to the accurate diagnosis of OSD. Every patient should undergo an evaluation of the tear film, eyelids, and lid margins. Traditional diagnostics such as tear breakup time, lid expression, and corneal/conjunctival staining help to identify the extent of disease and to distinguish between the aqueous-deficient and evaporative forms of dry eye disease.
The consequences of not treating OSD are myriad. A compromised ocular surface can adversely affect corneal imaging, keratometric readings, and IOL power calculations. OSD may also delay healing and cause patients to be unhappy with their surgical outcomes. Surgeons’ awareness and knowledge of corneal imaging are essential to making informed decisions, obtaining optimal results, and avoiding complications. n
1. Baudouin C, De Lunardo C. Short-term comparative study of topical 2% carteolol with and without benzalkonium chloride in healthy volunteers. Br J Ophthalmol. 1998;82(1):39-42.
2. Bell NP, Felman RM, Zou Y, et al. New technology for examining the anterior segment by ultrasonic biomicroscopy. J Cataract Refract Surg. 2008;34(1):121-125.
3. Ishikawa H, Schuman JS. Anterior segment imaging: ultrasound biomicroscopy. Ophthalmol Clin North Am. 2004;17(1):7-20.
4. Kahook MY, Noecker R. Quantitiative analysis of conjunctival goblet cells after chronic application of topical drops. Adv Ther. 2008;25(8):743-751.
5. Leung EW, Medeiros FA, Weinreb RN. Prevalence of ocular surface disease in glaucoma patients. J Glaucoma. 2008;17(5):350-355.
6. Lemp MA. Report of the National Eye Institute/Industry workshop on clinical trials in dry eyes. CLAO J. 1995;21(4):221-231.
7. Schaumberg DA, Sullivan DA, Buring JE, Dana MR. Prevalence of dry eye syndrome among US women. Am J Ophthalmol. 2003;136(2):318-326.
8. Schaumberg DA, Buring JE, Sullivan DA, Dana MR. Hormone replacement therapy and dry eye syndrome. JAMA. 2001;286(17):2114-2119.
9. Schaumberg DA, Sullivan DA, Dana MR. Epidemiology of dry eye syndrome. Adv Exp Med Biol. 2002;506(pt B):989-998.
10. Schein OD, Hochberg MD, Muñoz B, et al. Dry eye and dry mouth in the elderly: a population-based assessment. Arch Intern Med. 1999;159(12):1359-1363.
11. The epidemiology of dry eye disease: report of the Epidemiology Subcommittee of the International Dry Eye WorkShop (2007). Ocul Surf. 2007;5(2):93-107.
12. Ursea R, Silverman R. Anterior segment imaging for the assessment of glaucoma. Expert Rev Ophthalmol. 20105(1):59-74.
13. Yee RW. The effect of drop vehicle on the efficacy and side effects of topical glaucoma therapy: a review. Curr Opin Ophthalmol. 2007;18(2):134-139.
14. Reinstein DZ, Archer TJ, Gobbe M, et al. Stromal thickness in the normal cornea: three-dimensional display with Artemis very high-frequency digital ultrasound. J Refract Surg. 2009;25(9):776-786.
15. Reinstein DZ, Gobbe M, Archer TJ, et al. Epithelial, stromal, and total corneal thickness in keratoconus: three-dimensional display with Artemis very high-frequency digital ultrasound. J Refract Surg. 2010;26(4):259-271.
16. Silverman RH, Urs R, Roychoudhury A, et al. Epithelial remodeling as basis for machine-based identification of keratoconus. Invest Ophthalmol Vis Sci. 2014;55(3):1580-1587.
Alice T. Epitropoulos, MD
• cofounder and owner, The Eye Center of Columbus in Columbus, Ohio
• partner, Ophthalmic Surgeons & Consultants of Ohio, Columbus, Ohio
• clinical assistant professor, Department of Ophthalmology,The Ohio State University Wexner Medical Center, Columbus, Ohio
• (614) 221-7464; aepitrop@columbus.rr.com
• financial disclosure: consultant to TearScience
