CASE PRESENTATION
An 84-year-old woman of European ancestry presents with advanced to end-stage chronic angle-closure glaucoma. The patient underwent trabeculectomy with adjunctive mitomycin C 1 year ago in the right eye and 2 years ago in the left eye. She subsequently underwent cataract surgery, but the anterior chamber in each eye remained narrow with a maximal central depth of less than two central corneal thicknesses. Despite optically closed angles, IOP remained well controlled in the low teens in each eye until 3 months ago. Two weeks before the current visit, the IOP increased to more than 40 mm Hg in the left eye, and the patient began treatment with latanoprost, a fixed combination of dorzolamide and timolol, and brimonidine.
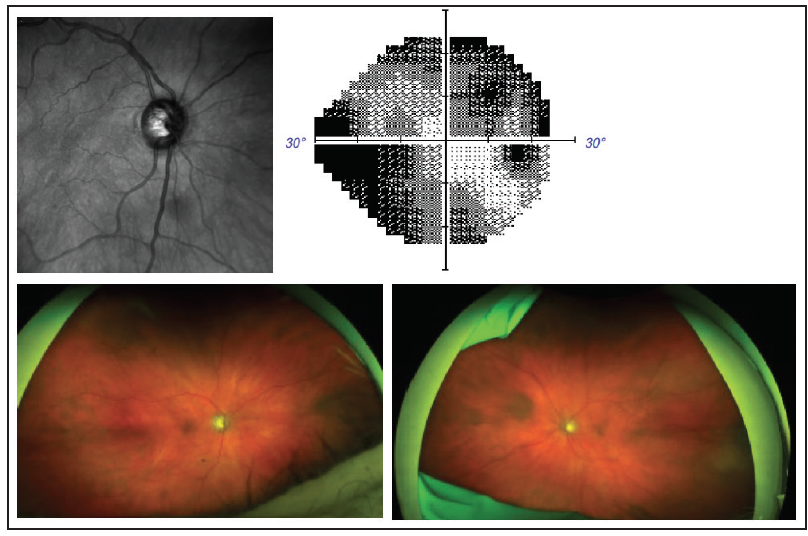
Figure. Topographic reconstruction of the optic nerve head of the right eye from OCT (top left). Achromatic automated visual field test of the right eye (top right). Widefield fundus camera image with peripheral artifact (bottom right). Widefield fundus camera image with inferior lid artifact (bottom left). On examination, the peripheral retina was thought to be normal in each eye.
At the current visit, visual acuity is 20/70 OD and counting fingers temporally in the left eye. IOP is 13 mm Hg OD and 34 mm Hg OS. Central corneal thickness measures 553 µm OD and 554 µm OS. A slit-lamp examination of the right eye shows a clear cornea with 2+ guttata; a superior, low (ie, height of approximately one corneal thickness), diffuse bleb with minimal vascularization; and a shallow anterior chamber cornea-to-IOL depth (ie, a height of approximately two corneal thicknesses). In the left eye, the cornea is clear with 1+ guttata and 360º of obvious iridocorneal touch with a cornea-to-IOL depth of approximately one corneal thickness. The IOL is in the bag in each eye.
The cup-to-disc ratio is 0.85 in the right eye and 0.95 in the left eye. Visual field testing shows dense superior and inferior arcuate defects in the right eye; testing is not possible in the left eye because of a loss of central fixation. Optic nerve reconstruction from OCT shows significant cupping in the right eye, whereas an image cannot be obtained in the left eye because of a small pupil. The peripheral retina in each eye is normal (Figure).
How would you proceed?
—Case prepared by Douglas J. Rhee, MD
THOMAS PATRIANAKOS, MD
This challenging case requires a stepwise approach to both diagnosis and management. The pressure spike might have been caused by a pupillary block component or by secondary angle closure without pupillary block from anterior rotation of the ciliary body–lens complex. Pupillary block is a less likely cause because the eye is pseudophakic and underwent a trabeculectomy, presumably with an iridectomy. Caution is warranted, however, because the site of an iridectomy can develop scarring or a fibrous membrane that can obstruct the flow of aqueous between the two chambers. If any component of pupillary block is suspected, a laser iridotomy should be attempted.
Ultrasound biomicroscopy could help to identify the etiology of the problem. Anteriorly rotated ciliary processes can arise from aqueous misdirection or from extreme zonular laxity such as that seen with exfoliation syndrome. Medical treatment with atropine and mannitol could be initiated to push the lens-iris diaphragm posteriorly and open the angle. If medical treatment does not work, treatment with an Nd:YAG laser can be attempted through the iridectomy to break the anterior hyaloid face and make the eye unicameral. A lensectomy and anterior hyaloid zonulectomy may be required for definitive treatment. An angle-based glaucoma procedure such as a trabeculotomy or goniosynechiolysis could be combined with one of the aforementioned options if the angle has been crowded for longer than 6 months, which would have increased the likelihood of irreversible damage to the trabecular meshwork.
ANGELA TURALBA, MD
This patient with primary angle-closure glaucoma whose IOP was previously controlled after trabeculectomy presents with an acute IOP elevation in the left eye. In this situation, gonioscopy is helpful for determining if the sclerostomy from the prior trabeculectomy is patent. When peripheral anterior synechiae develop in the area of the sclerostomy, it can be opened either by using an Nd:YAG laser to incise the iris or an argon laser to pull the iris away from the sclerostomy.
If the sclerostomy appears to be patent or cannot be visualized because of narrowing of the angle, it may be that the patient is experiencing acute and chronic aqueous misdirection syndrome. Features consistent with this picture would be central shallowing of the anterior chamber and a normal funduscopic examination. If this is the case, medical treatment with aqueous suppressants and atropine may break the attack.
If, however, the IOP remains high, surgical treatment may be warranted. Given the poor visual potential of the eye and the risks of intra- and postoperative complications with incisional surgery, transscleral diode cyclophotocoagulation would be a reasonable option for lowering the IOP. If the eye had better visual potential, an alternative would be to create a unicameral eye by performing pars plana vitrectomy, zonulectomy, and iridectomy. Another option would be to implant a pars plana tube if there is a high risk of persistently high IOP and peripheral anterior synechiae have closed the angle completely.
WHAT I DID: DOUGLAS J. RHEE, MD
A rho-kinase inhibitor would not be expected to work in an eye with angle closure. This patient did not want to undergo a pars plana vitrectomy and placement of a glaucoma drainage device in the pars plana. There was no anatomic space in the anterior or posterior chamber in which to place a tube. We discussed the option of cyclodestruction, and she elected to undergo micropulse diode cyclophotocoagulation in the left eye.
One day after surgery, visual acuity in that eye was light perception, and IOP was 27 mm Hg. The clinical examination was unchanged. I instructed the patient to resume therapy with antiglaucoma medications and start administering prednisolone acetate, and she was scheduled to return for follow-up in 2 days.



