Husam Ansari, MD, PhD
The COVID-19 pandemic has presented several insights into my practice and my patients that I had either forgotten or not yet realized.
At the peak of the pandemic, when we were only allowed to see urgent cases, I adopted video-based telemedicine visits with patients. Obviously, I could not conduct any meaningful eye examination for glaucoma purposes, but the ability to speak with patients without time pressure, to talk about their quality of life, to discuss their medication use, and to see inside their homes was extremely gratifying. The gratitude those patients showed in return made me realize how much a physician's attention matters to them. Patients appreciate the check-in, even if nothing practical (eg, IOP measurements) can be accomplished. I never realized before how much they relied on my reassurance.
Something else the pandemic has highlighted for me is the brazen practice we had of making our patients wait for hours in the office during their appointments. I used to make no apologies for this. The pandemic, however, has made it obvious how inappropriate that is. I am excited that my practice has “cracked the code” and discovered a way to get our patients in and out quickly and safely while still delivering outstanding care.
Larissa Camejo, MD
Through the COVID-19 pandemic, I learned that physicians will always find a way to provide care to their patients. Telemedicine has been a topic of discussion for years without reaching implementation. Although it continues to have significant limitations for our field, telemedicine certainly served a purpose at the height of COVID-19 closures.
I found the practice of telemedicine to be particularly and surprisingly useful with new patients. Some individuals had been waiting a few weeks for a new patient appointment with our practice, only to have that appointment canceled due to COVID-19. Most of our new glaucoma evaluation patients have undergone fairly recent examinations and testing by their referring ophthalmologists. Therefore, the telemedicine appointments allowed me to meet these patients, evaluate current available data, discuss their glaucoma status and potential next steps (pending confirmation on examination), and answer any questions they had. Many of these new patients are told that they are being referred because their glaucoma is advancing and may require surgery. Therefore, the telemedicine visits seemed to have a reassuring effect on these individuals. Many expressed gratitude to my team and me for holding these appointments.
Additionally, we were able to combine telemedicine appointments with quick IOP checks or testing in the office when needed. When we reopened, this practice allowed us to hit the ground running.
In terms of glaucoma management, it became obvious during the pandemic that there is a true need for an affordable way for patients to measure their IOP at home. Glaucoma care has seen so much innovation in the past few years that I wonder if an all-inclusive diagnostic machine, instrument, or application could become a reality in the future.
Xiongfei Liu, MD
At the heart of interventional glaucoma is the concept of individualized treatment that mixes and matches medication, laser, and surgical options that are tailored to each patient. It is a fluid concept that has been fueled by the challenges brought forth by the COVID-19 pandemic.
During the early phase of COVID-19’s impact, the volume of our clinical visits was restricted mainly to urgent cases. Unless the cause of elevated IOP was related to active neovascular or uveitic glaucoma, I performed more Xen Gel Stent (Allergan) surgeries in lieu of trabeculectomy and glaucoma drainage device procedures because Xen implantation is faster, has a better safety profile, and has a less-intensive postoperative course than trabeculectomy. For patients with the appropriate anatomy, postoperative visits can be minimal. At the height of the pandemic, the goals of surgery were to be fast, to be efficient, and to bring the patient’s IOP to a reasonable level, and Xen exemplified that. Furthermore, during this time, I used Ahmed Glaucoma Valves (New World Medical) rather than Baerveldt glaucoma implants (Johnson & Johnson Vision) to control IOP because fenestration can bring unpredictable IOPs in the first few postoperative weeks.
In California where I practice, the easing of quarantine by the end of May allowed more patients (by their choice) to follow up in clinic while adhering to multiple safety measures in place. However, social distancing restrictions meant limited space and limited staff. In July, the availability of Humphrey visual field (HVF, Carl Zeiss Meditec) testing was overwhelmed as more patients returned to the office while following the safety measures. One solution was to triage some patients to undergo visual field testing with the frequency doubling technology Humphrey Matrix (Carl Zeiss Meditec) instead. These patients tended to include those whose IOPs and visual fields were stable over the past few years. Although the interpretation is different from the HVF due to different mechanisms of measurement, I think that the Humphrey Matrix device gets the job done for carefully selected patients.
As the volume of clinical visits returned to equilibrium, I actively gauged each patient’s comfort level regarding therapy in light of COVID-19. Although some were conservative and preferred a minimal approach, others were quite comfortable with intervention (especially after they met the yearly deductibles). Depending on each patient’s circumstances, I backed off somewhat if patients had several medical comorbidities of old age, if they had a conservative attitude toward COVID-19, and if their disease progression was slow. For others, interventions continued with a few adjustments.
The arrival of the bimatoprost implant (Durysta, Allergan) helped to minimize clinical visits in a couple of situations. One scenario was for patients with adherence issues and/or difficulty administering their eye drops. Another scenario was for ocular surface improvement in preparation for Xen implantation; an improved ocular surface likely improves outcomes with the Xen and results in less follow-up.
For patients with cataract symptoms on multiple glaucoma drops, my preference has not changed with the trabecular bypass device. However, for canaloplasty I preferred the new handle for the Omni Surgical System (Sight Sciences) that came out this year. When combining goniotomy with canaloplasty, I left a couple of quadrants of the trabecular meshwork behind, even in cases of advanced glaucoma, in order to augment the effect of netarsudil/latanoprost ophthalmic solution (Rocklatan, Aerie Pharmaceuticals) in the event of a pressure spike and to potentially minimize the need for office visits to titrate medications.
Another consideration I kept in mind while treating patients was the COVID-19 situation in 2021. With vaccination on the horizon, the situation will likely improve, with more patients wanting surgical therapy. Furthermore, the Preserflo MicroShunt (Santen) will likely become available in the United States in 2021, which gives me more confidence to discuss the Xen with pseudophakic patients now, because the Preserflo device can be used in cases in which the Xen is not successful.
In terms of clinic flow, our practice has effectively utilized telemedicine. Because HVF slots are limited on the day of clinic visits, some patients undergo HVF/imaging visits with an IOP check by well-trained technicians. If the patient’s IOP is within a couple of points from baseline IOP, then they may return home. A couple weeks later, they will have a telehealth visit with their provider to discuss their imaging results and the plan moving forward. These telehealth visits can be intermixed with regular clinic visits to improve the efficiency of clinic flow (Figure 1).
Overall, 2020 has been a challenging year that will have a lasting impact. Despite these challenges, many promising solutions have emerged. At its heart, interventional glaucoma will continually evolve and adapt to the situation at hand so that 2021 can be brighter for glaucoma patients.
Michael D. Greenwood, MD
As the saying goes, necessity is the mother of invention. With the changes introduced to our practice patterns to minimize patient visits and contact during the COVID-19 pandemic, that certainly seemed to be the case this year.
During the pandemic, I have learned that we need more options for sustained drug delivery and more methods of monitoring patients with glaucoma outside the clinic. Currently, one sustained drug delivery option is available, but access to an option that is longer-lasting or repeatable would go a long way for patients.
As for monitoring patients outside the clinic, we simply need more available solutions, whether it be a take-home tonometer, an implantable IOP-monitoring device, or perhaps even an automated method similar to the blood pressure cuffs located in many stores and pharmacies. Diagnostic devices such as these could provide physicians with substantially more information about their patients’ conditions and could alleviate patients’ anxiety about their disease and/or access to their physicians.
Shakeel Shareef, MD
The COVID-19 pandemic brought to light the importance of education, communication, empathy, and reassurance. It also emphasized the need to remind patients of the chronicity of glaucoma and of its nature as a slowly progressive neurodegenerative disease. Glaucoma specialists play an important role in the doctor-patient relationship to allay patients’ fear of going blind. Irrespective of disease severity, several clinical trials spanning the disease spectrum from ocular hypertension to advanced disease enable me to counsel patients that, in general, glaucoma can be kept under control.
During the temporary ban on elective surgeries due to COVID-19, glaucoma management took on a new meaning for nonurgent cases. Optimization of medical therapy in my practice was twofold. First, we utilized fixed-combination medications to enhance compliance. Second, we emphasized the role of proper drop instillation with either eyelid closure or nasolacrimal occlusion1 to increase ocular absorption and drug efficacy while minimizing systemic absorption to avert side effects.
The slogan “an educated consumer is our best customer” pays dividends by placing the onus on patients to take their drops properly. I remind patients, “It’s not what you take but how you take it.” For elderly individuals who live alone or for those with involuntary hand tremors or arthritis, nasolacrimal occlusion may be impractical. Given the challenge of eye-hand coordination in properly placing an eye drop, I simply ask patients to close their eyelids for 3 to 5 minutes. This approach is particularly important in patients with thick corneas on pachymetry. Looking at a given patient’s IOP clinical data spanning several years, I have observed a sustained reduction in IOP of as much as 4 to 5 mm Hg not seen in prior visits. For patients who are intolerant of multiple glaucoma medications or who quickly run out of their drops due to excessive bottle squeezing, I have offered preservative-free alternatives, necessitating the use of individual vials for each drop not only to enhance tolerability but also to avoid waste, respectively.
Given the negative impact that COVID-19 has had on the economy, the findings of the Selective Laser Trabeculoplasty Versus Eye Drops for the First-Line Treatment of Ocular Hypertension and Glaucoma (LIGHT) trial2 seem especially relevant. In this trial, treatment-naïve patients with open-angle glaucoma or ocular hypertension were randomly assigned to receive selective laser trabeculoplasty (SLT) or eye drops as a first-line treatment. According to the 3-year data, more patients in the SLT arm than the eye drop arm achieved their target IOP (93% vs 91.3%), and SLT was more cost-effective than medical therapy. Additionally, none of the SLT patients required surgical intervention during the study period, compared with 11 patients in the eye drop group. I now offer patients the option of laser treatment either as first-line therapy or earlier in the treatment paradigm instead of escalating medical therapy to control IOP.
For patients who require surgical intervention, the pandemic has emphasized the importance of being patient-centric when selecting a procedure. This entails minimizing the frequency of postoperative visits while achieving an acceptable level of IOP. Even before COVID-19, a significant trend among Medicare recipients has been observed in which surgeons are moving away from performing primary trabeculectomy3 (and its labor-intensive postoperative period) and toward nonbleb-forming procedures such as MIGS. Schlemm canal–based procedures offer a built-in safety net against hypotony due to the episcleral venous pressure, especially in elderly patients. From a quality-of-life standpoint, I offer MIGS to patients who are administering multiple medications by striking a balance between safety and a reduction in postoperative visits, with the compromise of still having to instill glaucoma medications, albeit with a reduced postoperative treatment burden.
A report from the AAO showed that, among patients with medically controlled glaucoma who needed cataract surgery and had not undergone prior incisional surgery, cataract surgery alone resulted in a sustained IOP reduction out to 1.5 to 3 years in those with primary open-angle glaucoma (13%), pseudoexfoliative glaucoma (20%), and chronic angle-closure glaucoma (30%).4 Patients with chronic angle-closure glaucoma were found to have an additional 58% decrease in medication burden. During the pandemic, I have offered select patients the option of cataract surgery as a standalone procedure, with the understanding that they may need additional glaucoma intervention in the future. This approach provides peace of mind to both the doctor and the patient that, until the pandemic is brought under control with the distribution of FDA-approved vaccines, cataract surgery alone also plays a role akin to a MIGS procedure in lowering and controlling IOP.
CASE EXAMPLE
I recently treated a 92-year-old patient who was administering four glaucoma medications with a visual acuity of 20/400, an IOP of 20 mm Hg OD, a mature age-related nuclear sclerotic cataract, and advanced cupping. She had progressive visual field loss with a pitch-black field noted in the right eye on a 24-2 Humphrey Field Analyzer test (Carl Zeiss Meditec; Figure 2). Kinetic perimetry of the right eye (Figure 3) revealed an inferior island of vision confined to within 30º with split fixation. Although an ab externo procedure was an option to lower her IOP, given her limited mobility, dependence on others for transportation, and very thin bulbar conjunctiva on examination, I was concerned about her risk of perioperative suprachoroidal hemorrhage, the need for intensive postoperative medical therapy, and multiple office visits during the pandemic.
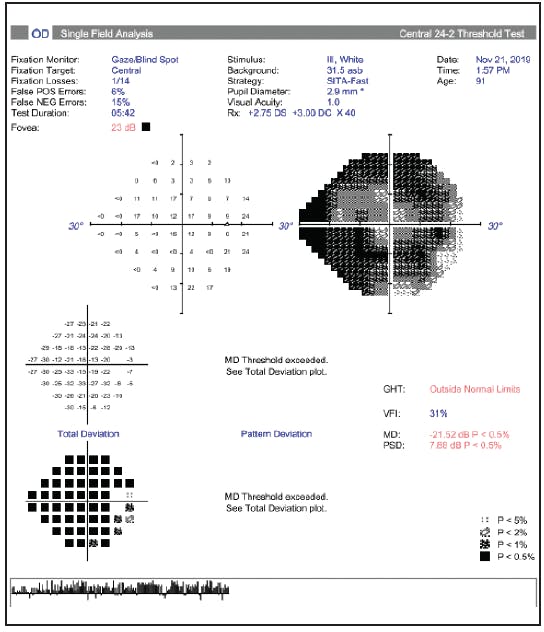
Figure 2. Humphrey Field Analyzer with Swedish Interactive Threshold Algorithm Fast testing showed generalized diffuse depression.

Figure 3. A kinetic Social Security Administration test was performed with the Humphrey Field Analyzer 3. The test uses preset parameters of III 4e (size and intensity), 4º/sec (speed), and white (color) background and identifies the farthest perimeter of the visual field at eight meridians located 45º apart.
As a result, I took a patient-centric approach and considered a Schlemm canal–based procedure, while keeping in mind that episcleral venous pressure functions to counter hypotony, which can be associated with a risk of choroidal expansion. The 3-year findings of the HORIZON trial showed no difference in corneal endothelial cell loss between patients randomly assigned to undergo combined phacoemulsification and Hydrus Microstent (Ivantis) implantation versus cataract surgery alone.5 This finding was important in my efforts to minimize endothelial cell loss in my nonagenarian patient, not only during surgical manipulation but also in the postoperative period long-term. Further, the HORIZON study showed that 73% of eyes in the combined phacoemulsification-Hydrus group were medication-free, compared with 48% in the cataract surgery–only group. Given the life expectancy of my patient, I felt that this was a reasonable compromise to help her maintain her level of visual functioning for the remainder of her life. The HORIZON study also showed that patients in the combined group had a lower likelihood of IOP spikes greater than 40 mm Hg on postoperative day 1 than patients in the cataract surgery–only group (1.4% vs 14.4%).6 Additionally, the incidence of IOP at least 10 mm Hg above baseline on postoperative day 1 was significantly lower (3.0% vs 22.5%). This is critical in a patient with split fixation at risk of a snuff-out of central vision.7
The patient underwent combined phacoemulsification and Hydrus surgery under topical anesthesia. At her last postoperative visit, visual acuity was 20/80, and IOP was 11 mm Hg on one glaucoma medication. Incorporating well-designed studies into my practice and taking a patient-centric approach in each case has enabled me not only to minimize postoperative complications and visits but also to help patients maintain their quality of life and avert blindness.
1. Flach AJ. The importance of eyelid closure and nasolacrimal occlusion following the ocular instillation of topical glaucoma medications and the need for the universal inclusion of one of these techniques in all patient treatments and clinical studies. Trans Am Ophthalmol Soc. 2008;106;138-145.
2. Gazzard G, Konstantakopoulou E, Garway-Heath D, et al. Selective laser trabeculoplasty versus eye drops for first-line treatment of ocular hypertension and glaucoma (LiGHT): a multicenter randomized controlled trial. Lancet. 2019;393(10180):1505-1516.
3. Arora KS, Robin AL, Corcoran KJ, Corcoran SL, Ramulu PY. Use of various glaucoma surgeries and procedures in Medicare beneficiaries from 1994-2012. Ophthalmology. 2015;122:1615-1624.
4. Chen PP, Lin SC, Junk AK, et al. The effect of phacoemulsification on IOP in glaucoma patients: a report by the American Academy of Ophthalmology. Ophthalmology. 2015;122(7):1294-1307.
5. Ahmed IIK, Rhee DJ, Jones J, et al. Three-year findings of the HORIZON trial: a Schlemm canal microstent for pressure reduction in primary open angle glaucoma and cataract. November 5, 2020. Ophthalmology. doi:10.1016/j.ophtha.2020.11.004
6. Zebardast N, Zheng C, Jampel H, et al. Effect of a Schlemm’s canal microstent on early postoperative intraocular pressure after cataract surgery: an analysis of the HORIZON randomized controlled trial. Ophthalmology. 2020;127(10):1303-1310.
7. Kolker AE. Visual prognosis in advanced glaucoma: a comparison of medical and surgical therapy for retention of vision in 101 eyes with advanced glaucoma. Trans Am Ophthalmol Soc. 1977;75:539-555.
John T. Lind, MD, MS
COVID-19 has been a disruptive force in patient care during 2020, but not all disruptive forces are bad. We have learned how to connect to patients outside our brick-and-mortar clinics. We have learned how to rely on our colleagues’ records and purpose new therapies without a traditional face-to-face encounter. We have learned how to stress compliance and address patients’ needs with a phone call or virtual visit. We have learned new operation protocols that have been developed out of necessity to make care safer for patients and staff.
Alena Reznik, MD
During the COVID-19 pandemic, my colleagues and I have scrambled to adjust follow-up care by extending follow-up visits for stable patients and prioritizing our unstable patients. We have changed our office appointment templates to prevent patient overlap in the waiting area, and cleaning protocols were scrutinized and reenforced.
Additionally, we implemented the use of telemedicine for stable patients to emphasize medication compliance; however, this practice lacks the capability to conduct an IOP check and optic nerve examination. The COVID-19 pandemic has emphasized the need for at-home IOP measurement in an affordable manner because insurance coverage is currently lacking.
Oluwatosin Smith, MD
These have certainly been trying times for physicians and patients alike. The COVID-19 pandemic has revealed both the willingness of physicians and patients to adapt and the resilience of both parties when faced with a difficult situation. It has also highlighted the concerns many patients have regarding their chronic, potentially blinding eye condition.
My insight into the disease state itself has not changed much in the COVID-19 era; what has become more apparent is our major dependence on office visits to manage, in a snapshot fashion, a disease with a labile, treatable risk factor. We have struggled to get patients back into the office for their “snapshot visit” and have had to adapt our clinic routines to get the most basic or minimal evaluation in the form of visual acuity and IOP checks done in a safe and efficient manner. In turn, this has caused us to start thinking more about ways of monitoring patients at home or in other virtual ways.
In light of the pandemic, we have seen greater consideration of the optimization of telemedicine, home tonometers for IOP monitoring, and visual field testing, to name a few. These solutions might have helped us better sort out the patients with a critical need for follow-up if they had been part of our routines prior to the outbreak of COVID-19. Additionally, the use of multimonth medication refills has become preferred, as have surgical options that require less follow-up and have better safety profiles.
It has been interesting to see the variation in what has been of most concern to patients during this time. Loss of vision and the ability to refill their medications certainly seem to rank high on their list of concerns.
Daniel Lee, MD
In the early months of the COVID-19 pandemic, my colleagues and I made our best efforts to triage patients. Calculated risks were taken, and patients who were deemed high risk were kept on the schedule while others' appointments were postponed indefinitely.
As our “lower-risk” patient population began to filter back into the practice, several had unexpectedly elevated IOPs and optic nerve progression despite long track records of stability. On the other hand, some patients with “high-risk and uncontrolled” glaucoma who chose to self-quarantine and cancel their visits returned with stable disease.
The pandemic has served as a reminder that the course of glaucoma can be nonlinear and unpredictable. Our best answer to this unpredictable nature is close and frequent monitoring. This experience has highlighted our need for at-home testing, and I look forward to the many innovations in telehealth technology that are under way.










