CASE PRESENTATION
In the May/June 2021 installment of this column, I presented the case of a 67-year-old woman who developed recurrent hyphemas in the right eye. The patient had undergone uneventful cataract surgery and placement of a Hydrus Microstent (Alcon) in each eye 2 years earlier and had a remote history of a scarred trabeculectomy in the right eye.
Although the IOP remained well controlled without medication, 18 months after cataract surgery and MIGS, she began experiencing recurrent hyphemas in the right eye. The patient was administering no glaucoma medication; she is allergic to benzalkonium chloride and cannot tolerate most eye drops. She had started therapy with clopidogrel bisulfate (Plavix, Bristol-Myers Squibb and Sanofi Pharmaceuticals) a year earlier to treat coronary artery disease.
The Hydrus was removed because it appeared to be localized next to an aberrant vessel and repositioned inferiorly to help with IOP control. In the weeks following surgery, the patient was free of hyphema and glaucoma medication. (Click here to read the original case presentation in its entirety.)
Seven months after the stent was repositioned, the patient presents with sudden pain and hazy vision in the right eye. She is not administering any glaucoma medication. Her UCVA has decreased from 20/30 to hand motions OD and is stable at 20/20 OS. The IOP is 31 mm Hg OD and 18 mm Hg OS. A slit-lamp examination reveals a scarred trabeculectomy and 1-mm hyphema in the right eye and a well-positioned IOL in each eye. On gonioscopy, the appearance of the stent in the inferior angle of the right eye is unremarkable, but bleeding is evident at the site where the device was previously removed (Figure 1). A hazy view in the right eye notwithstanding, a fundus examination reveals cupping of both optic nerves. Previous visual field testing demonstrated stable superior arcuate scotomas in both eyes, and OCT imaging shows cupping and thinning of the retinal nerve fiber layer in both eyes (Figures 2 and 3).

Figure 1. A gonioscopic view of the nasal angle shows the site of previous stent removal and emanating hyphema from a break in Schlemm canal spanning less than 1 clock hour.
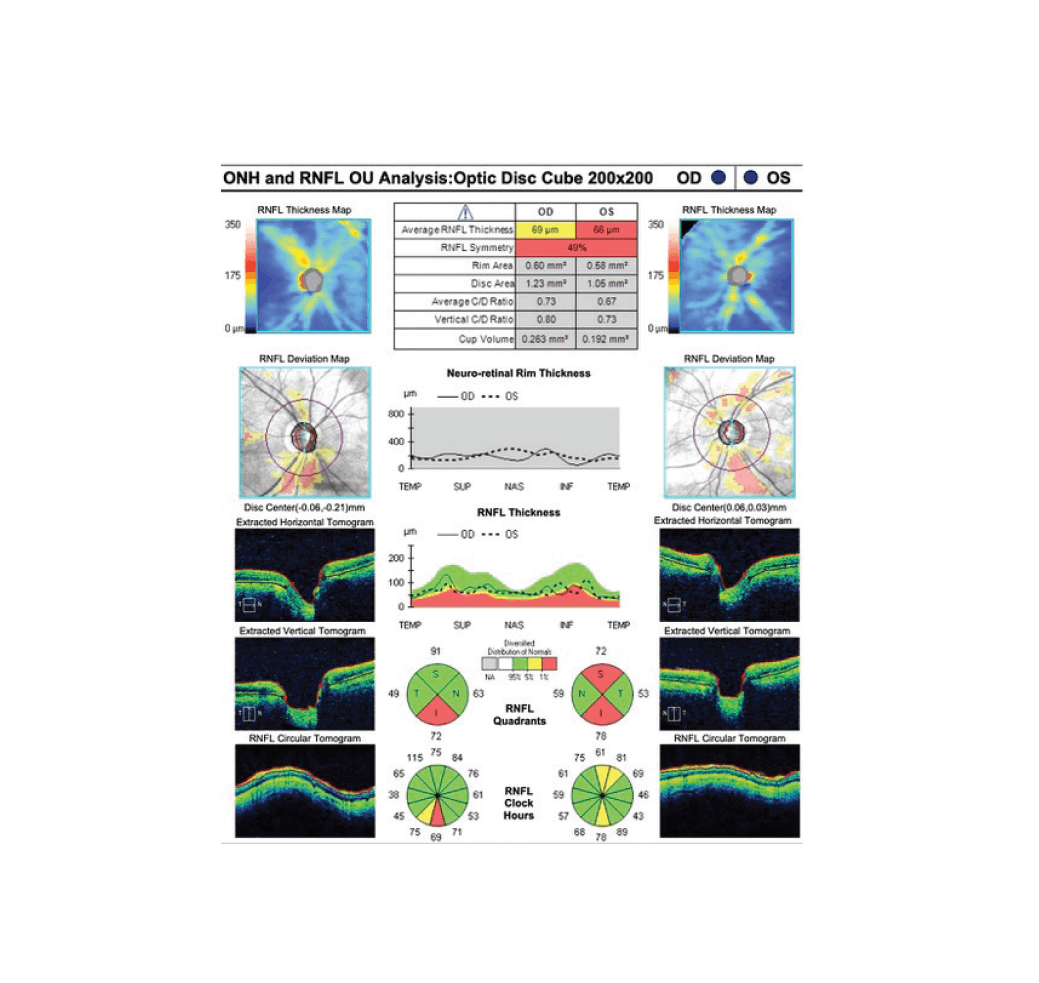
Figure 3. OCT scan shows cupping of the optic nerve and thinning of the retinal nerve fiber layer in each eye.
The patient is exasperated that the hyphema has recurred and desperately wants a permanent solution to the problem. How would you treat the hyphema? How would you manage her IOP, given the difficulty she has tolerating medication and her desire to continue avoiding IOP-lowering medical therapy?
—Case prepared by Jacob Brubaker, MD
SAHAR BEDROOD, MD, PHD
The removal and inferior placement of the Hydrus worked for a short period of time. Given the hyphema’s recurrence in the original area of the device’s placement, there is likely blood reflux from exposed blood vessels in Schlemm canal. The delicate blood vessels in combination with the use of an anticoagulant puts the patient at risk of recurrent hyphema.
One option for addressing the recurrent bleeding is to cauterize the nasal scleral blood vessels that are part of the downstream network of blood vessels that take blood away from the angle. Cautery in that area may reduce the blood reflux and stop the recurrent hyphemas.
The patient clearly cannot tolerate angle surgery or a device placed in the angle. The angle and iris root are laden with blood vessels, and no intervention to lower IOP should disrupt or rub against them because of the anticoagulant therapy. The current IOP spike is likely due to the hyphema. Given the advanced visual field defects and her intolerance of topical glaucoma drops, I would consider placing either a Xen Gel Stent (Allergan) or a tube shunt at the time of cautery to the scleral vessels to provide additional IOP lowering.
XIONGFEI LIU, MD
The twists and turns of this interesting case continue. The recent hyphema occurred at the site of previous Hydrus implantation. The bleeding could be the result of a break in Schlemm canal; with blood reflux into the canal, red blood cells can trickle into the anterior chamber. A fundus examination should be performed to rule out retinal pathology if the view is not obscured.
To control IOP in the short term, therapy with a preservative-free aqueous suppressant, a cycloplegic, and a steroid would be initiated. If the patient is a potential steroid responder, loteprednol etabonate ophthalmic ointment 0.5% (Lotemax, Bausch + Lomb) with a fast taper would be prescribed instead of prednisolone acetate 1%. I would also recommend that the patient avoid certain body positioning and Valsalva maneuvers.
Preventing hyphema in the long term requires a discussion of additional surgical procedures with the patient. Normally, I would lyse any peripheral anterior synechiae (PAS), but localized PAS over the previous Hydrus entry site would potentially be beneficial here. After a trial of atropine to determine if PAS form from pupillary dilation, argon laser treatment would be performed to coagulate bleeding at the entry site and promote scarring. Cryotherapy is not an option, because it would cause too much collateral damage. If laser treatment is unsuccessful, then a modified iridodialysis repair could be performed to plug the bleeding site. Because only 1 hour of iatrogenic PAS is needed, IOP is unlikely to increase significantly enough postoperatively to necessitate additional glaucoma surgery. If glaucoma surgery is required, however, then the implantation of a glaucoma drainage device or treatment with the Micropulse P3 Glaucoma Device (Iridex) could be considered.
WHAT I DID: JACOB BRUBAKER, MD
This case speaks to the difficulty of treating recurrent hyphemas after angle-based surgery. On presentation, the patient and I discussed possible treatment options. We elected to treat the area with argon laser ablation in an attempt to cauterize the source. The treatment was successful for 4 months, at which point the hyphema recurred, and the IOP rose to 61 mm Hg.
I struggled to determine the best next step. The patient and I discussed her situation at length. Finally, I suggested surgical correction involving treatment akin to a cyclodialysis repair. I postulated that creating a localized PAS at the site of bleeding would allow iris tissue to plug the site of bleeding, thus preventing future hyphemas. I stressed to the patient that the pupil would likely have a slightly peaked appearance afterward but that the treatment, if successful, could be a permanent solution.
In the OR, using a gonioprism, I delineated the site of Schlemm canal bleeding by marking the area with a marking pen. Next, a 10-0 nylon suture on a needle was passed through the limbus, then the peripheral iris, and back out the limbus (Figure 4). The suture was tied, and the iris was approximated into the angle to create a PAS that covered the site of bleeding (Figure 5). (Click here to watch the procedure.)
The following day in clinic, the patient’s IOP was 5 mm Hg, her visual acuity was 20/400, and a slight wound leak was evident at the suture track. At 1 week, the leak had sealed, the IOP was 16 mm Hg, and her visual acuity was 20/25 (Figure 6).
At 6 months—the patient’s most recent follow-up visit—her visual acuity was 20/25, and the unmedicated IOP was 18 mm Hg. She had experienced no further hyphemas and was extremely happy. Because the IOP was borderline, we discussed starting a topical glaucoma medication. The patient elected to follow up with her referring doctor close to home in 4 months and to add latanoprost ophthalmic emulsion 0.005% (Xelpros, Sun Ophthalmics) at that time if the IOP remains in the upper teens.









