In early 2019, a 67-year-old woman was referred by an area ophthalmologist for combined cataract and glaucoma surgery on her left eye. Nine years earlier, the patient had undergone complicated cataract extraction and trabeculectomy on her right eye. The IOL had to be placed in the sulcus, and aqueous misdirection syndrome necessitated a pars plana vitrectomy and iridozonulohyaloidectomy (IZHV) shortly after surgery. The sulcus-fixated IOL dislocated partially into the anterior chamber with iris capture of the optic. She experienced chronic corneal edema and poor vision. Details of the patient’s prior surgery and procedural care were unclear on presentation. Limited visual field and OCT data were provided.
At the consultation, the patient’s UCVA was 20/400 OD and 20/80 OS, which did not improve in either eye with refraction or a pinhole test. A relative afferent pupillary defect was evident in the right eye. The IOP was 9 mm Hg OD and 36 mm Hg OS. Corneal thickness was 590 µm OD due to chronic corneal edema and 540 µm OS.
The patient’s drug regimen consisted of travoprost administered at every bedtime and timolol maleate 0.5% dosed every morning in the left eye only. She had a history of intolerance of multiple topical glaucoma medications. Moderate ocular surface disease was evident in each eye.
Decades earlier, she had undergone bilateral laser peripheral iridotomies (LPIs) for narrow angles. The LPI in each eye was patent on presentation. The superior bleb in the right eye appeared flat, but the historical IOPs provided (previous 2–3 years) ranged from 6 to 13 mm Hg OD on no medication and 19 to 28 mm Hg OS on the drop regimen described earlier.
An examination found iris capture of the optic in the right eye and a dense cataract (grade 4 using the Lens Opacification Classification System) in the left eye. Gonioscopy revealed some peripheral anterior synechiae (PAS) in the right eye, but the view was limited by corneal edema. No PAS were present in the left eye. The view to the posterior segment was poor in the right eye, but a small optic nerve with significant superior retinal nerve fiber layer (RNFL) thinning was observable. The posterior segment of the left eye was unremarkable except that the cup-to-disc ratio was 0.6, superior RNFL thinning was evident, and the optic nerve was small. Biometry identified axial hyperopia in both eyes and a thick lens in the left eye (Figure 1). OCT imaging confirmed bilateral RNFL loss and notably underestimated the cup-to-disc ratio in each eye, likely owing to patient movement and a poor signal (Figure 2). Visual field testing detected a center-involving inferior arcuate scotoma in the right eye and a peripheral inferior arcuate scotoma in the left eye (Figure 3).
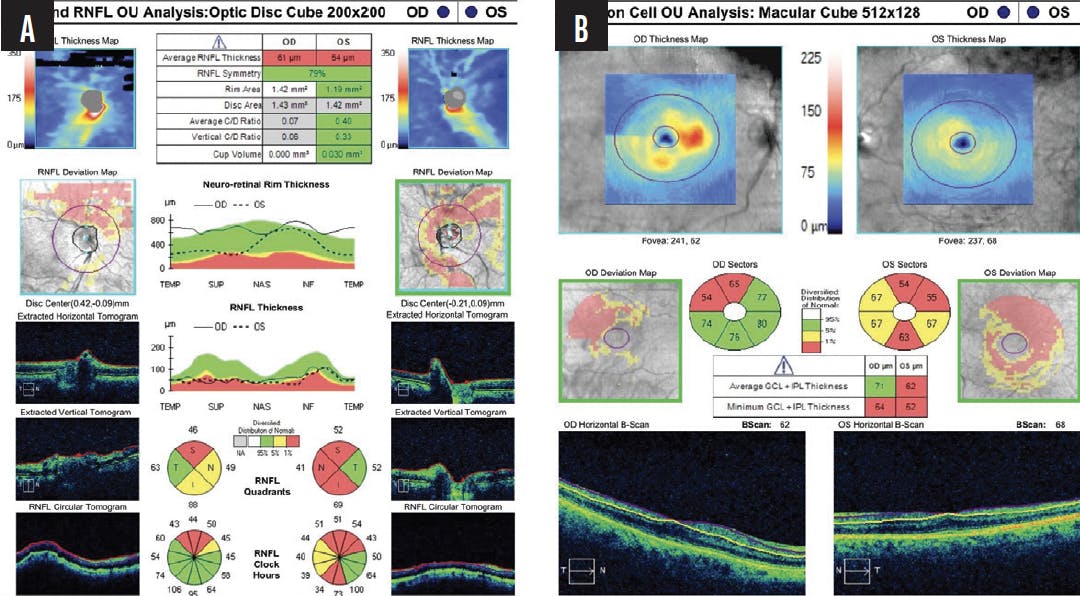
Figure 2. OCT analysis of the optic nerve head and RNFL (A) and ganglion cell analysis (B) of both eyes.
Despite the patient’s stated reluctance to consider surgery on the left eye, cataract removal to improve her vision and glaucoma surgery to improve IOP control were discussed. Less invasive options such as selective laser trabeculoplasty and additional or alternative topical medications were described, but it was noted that they would not improve her visual acuity and were unlikely to achieve the target IOP. It was explained that the patient’s surgical history and short axial length might have been the source of complications from cataract surgery in the right eye. Alternative angle-based and MIGS procedures that could be performed in combination with cataract surgery were discussed as options to reduce the risk of similar complications in the left eye. The patient said that she would discuss her situation with her husband and follow up with questions or concerns.
Given the surgical history of the patient’s fellow eye, how would you counsel her? What would your next step be in terms of IOP management?
—Case prepared by James Murphy, MD
ANNA T. DO, MD
The bilateral superior RNFL loss initially raises suspicion of superior segmental optic hypoplasia, especially because OCT imaging demonstrates thinness in the superonasal sector, which can correlate with inferior wedge-shaped field loss. The patient’s gonioscopy and nerve findings and elevated IOP, however, suggest that she has chronic angle-closure glaucoma in both eyes. Although the LPI in each eye is patent, the presence of elevated IOP in a relatively short eye with a 2.58-mm anterior chamber likely means there is persistent iridotrabecular meshwork contact.
The left eye has a visually significant cataract and requires a lower IOP. I would recommend phacoemulsification combined with transluminal viscodilation of Schlemm canal and goniotomy using the Omni Surgical System (Sight Sciences). The conventional outflow pathway may be compromised in an eye with chronic, progressive angle closure, but transluminal canal dilation—especially combined with cataract extraction—could still salvage trabecular outflow if no PAS are evident.
The history of aqueous misdirection in the right eye significantly increases the risk of this complication in the fellow eye. An IZHV, with the anterior vitrector placed through a clear corneal incision, would therefore be performed. I would try to avoid large IOP fluctuations during the perioperative period. Treatment with preoperative oral or intravenous acetazolamide would be administered to dehydrate the vitreous. Postoperatively, cycloplegic agents would be prescribed to help pull the zonules and lens diaphragm posteriorly.
MANJOOL SHAH, MD
The patient’s biometry measurements, examination findings, and interventional history suggest the primary angle-closure disease spectrum. The degree of optic neuropathy in the left eye is difficult to determine, and although it is likely glaucomatous, superior segmental optic nerve hypoplasia should be considered. With an IOP of 36 mm Hg, however, and evidence of angle closure, cataract extraction is the preferred intervention here based on the results of the Early Lens Extraction for the Treatment of Primary Angle-Closure Glaucoma (EAGLE) study.1
The density of the cataract seems to correspond to the patient’s BCVA, but the foveal threshold on visual field testing could help determine if visual recovery will be limited. Foveal threshold values should be obtained on visual field testing—at the very least for preoperative counseling in this situation.
Caution is warranted based on the history of malignant glaucoma (MG) in the patient’s fellow eye. A careful surgical technique would include maintenance of anterior chamber pressurization using either a dispersive OVD or balanced salt solution during the removal of automated irrigation. The goal would be to prevent transient anterior chamber depressurization with resultant choroidal expansion, which can incite MG. Although a history of MG in the patient’s right eye greatly increases the risk of complications in her left eye, an axial length of 21.6 mm makes it more likely that depressurization of the anterior chamber by the trabeculectomy was a greater contributor. I would plan to place a standard monofocal IOL in the capsular bag and orient the haptic away from the iridotomy in case a postoperative Nd:YAG IZHV is required.
Close monitoring of the postoperative refraction could flag anterior movement of the IOL, which would be suggestive of MG. Small eyes are generally at increased risk of a refractive surprise, but a significant and/or progressive myopic surprise suggests anteriorization of the IOL–capsular bag complex and requires intervention.
Given the patient’s monocular status, medication intolerance, chronic iridotrabecular contact, and resultant outflow dysfunction, I would typically recommend a canal-based intervention, although there is little evidence to support my inclination. A goniosynechialysis should be performed to open the angle maximally for 360º. An excisional goniotomy or off-label microstent implantation could be performed at the same time to help control the IOP while the trabecular meshwork dysfunction potentially abates.
ARSHAM SHEYBANI, MD
I suspect the gonioscopic findings in the left eye showed angle closure despite the patent LPI. Based only on the IOP, the patient would have qualified for clear lens extraction, but she also has primary angle-closure glaucoma. The visual field defect could be real, but it could also be a rim artifact. The ganglion cell analysis and OCT suggest that she likely has true visual field loss.
I would treat by mechanism and open the angle with cataract extraction. If there are PAS, I release them at the time of surgery. Without clear evidence of PAS, my preference is to add a 3–clock hour excisional goniotomy when patients have glaucomatous optic neuropathy. Given the patient’s intolerance of medication and the likely chronic nature of her angle closure, I am not sure the trabecular meshwork will be fully functional after the angle is opened. This is why I would add a goniotomy.
The frequency of MG increases in eyes with a short axial length, and the risk intensifies when it is shorter than 19 mm. I would not, however, perform a prophylactic IZHV here, because the axial length is greater than 21 mm and I suspect the filtration procedure precipitated the MG. Maintaining the anterior chamber during surgery and relying on the backstop of episcleral venous pressure would be safer options at this time.
Given the IOP, I would not delay surgery. I would prescribe an oral carbonic anhydrase inhibitor to reduce the IOP preoperatively. Large IOP swings would put the patient at risk of a suprachoroidal hemorrhage or could precipitate choroidal expansion.
AAKRITI GARG SHUKLA, MD
The patient's target IOP should be lower than her current measurements given her manifest damage and nearly monocular status. I recommend removing the lens to alleviate the angle-closure mechanism. Although I anticipate that lens extraction will have some beneficial effect on her IOP, she will likely need lower pressures to maintain vision in the long term.
A combined phacoemulsification and angle procedure would be my preference over filtering surgery, especially given her prior history. I would counsel the patient that she may need additional glaucoma surgery in the future and that we are planning on a staged approach for an optimal outcome. I would avoid a 360º gonioscopy-assisted transluminal trabeculotomy for this patient. The prevalence of hyphema after this procedure is high; given her monocular status, this could temporarily result in poor vision that is disabling. Instead, I would perform a 4-to-6–clock hour goniotomy with the Kahook Dual Blade (New World Medical), bent 25-gauge needle, or Omni Surgical System. If using the latter, I would perform canaloplasty as well as 180º goniotomy. These procedures are not recommended in the setting of blood thinner use; I would ask the patient about this prior to selecting an angle procedure. The placement of a trabecular microstent in the angle would be another alternative, but the indication for these devices is currently limited to open-angle glaucoma.
I typically reserve the use of IV mannitol 12.5 g to 25 g for patients with nanophthalmos; however, given the history of MG in the fellow eye, I would recommend administering this medication 1 hour before the case. Her surgical informed consent should include a possible IZHV if signs of MG are detected intraoperatively. Postoperatively, acetazolamide should be administered to prevent an overnight IOP spike, and the patient should be instructed to continue topical medications the night following surgery. She will need to be watched closely during the postoperative period for signs of MG.
WHAT I DID: JAMES MURPHY, MD
The patient elected to undergo cataract extraction combined with endocyclophotocoagulation, 360º canaloplasty, and goniotomy of the inferior 180º in the left eye. One day after surgery, her UCVA was 20/30, and the IOP was 12 mm Hg on a drug regimen of prednisolone acetate 1% four times per day and no glaucoma medication. One week postoperatively, her UCVA was 20/50, the IOP was 19 mm Hg, and the anterior chamber was shallower than immediately after surgery. Because of the patient’s history, the LPI was treated with an Nd:YAG laser, therapy with atropine 1% daily was initiated, and she was referred to a retina specialist. One week later, the anterior chamber remained shallow, and the IOP was 24 mm Hg. Her UCVA was 20/50, and her BCVA was 20/20 with a refraction of -1.00 D because of an anterior shift in the effective lens position of the posterior chamber IOL.
The patient’s preoperative regimen of glaucoma drops was restarted, and she was encouraged to undergo retinal surgery before the IOP became more difficult to control. An IZHV was performed within the week. Two weeks later, her BCVA was 20/20 with a refraction of +0.25 D, the anterior chamber was deep, and the IOP was 10 mm Hg. Therapy with timolol was halted. The patient was advised to continue administering travoprost hourly in the left eye and to taper the prednisolone over 3 weeks.
At the final postoperative visit, her UCVA was 20/20, and the IOP was 11 mm Hg on travoprost monotherapy, which was then discontinued. Three weeks later, her UCVA was 20/20, the IOP was 13 mm Hg on no medication, and a marked improvement in the signs and symptoms of ocular surface disease was noted.
Three years after surgery, the RNFL and visual fields were stable (Figures 4 and 5), the patient’s UCVA was 20/20, and the IOP ranged from 11 to 15 mm Hg. Because I have seen the IOP drift upward over time in similar patients, I informed her that, if the IOP ever exceeds 16 mm Hg at two or more consecutive visits or if RNFL loss or visual field progression is suspected, then it may become necessary to restart topical glaucoma therapy. Comanagement of the patient with her local ophthalmologist continues to the present day.

Figure 4. Visual field testing for the right (A and B) and left (C and D) eyes from 2019 until 2022 showed no evidence of disease progression.

Figure 5. OCT analysis of the RNFL (A) and ganglion cell analysis (B) in 2021 showed no disease progression.
This case illustrates a few important points regarding MIGS procedures. First, they can be powerful in terms of IOP lowering. Second, although MIGS is less invasive than traditional glaucoma surgery, serious postoperative complications can occur. Third, all glaucoma procedures are temporary solutions to a permanent condition. The last point guides my approach to patient care. When counseling patients, I describe a journey of maintaining vision and preventing glaucomatous progression that may involve several surgical steps. I find that, when patients understand that they may need multiple interventions over years or decades, they tend to be more at ease with and accepting of their individual journey.
1. Azuara-Blanco A, Burr J, Ramsay C, et al; EAGLE study group. Effectiveness of early lens extraction for the treatment of primary angle-closure glaucoma (EAGLE): a randomised controlled trial. Lancet. 2016;388(10052):1389-1397.









