AT A GLANCE
- The assessment of corneal biomechanics may come to play an important role in glaucoma management and risk stratification.
- Current methods of assessing corneal biomechanics are promising, but they are limited to some degree by the confounding effects of IOP.
- Future studies should take into account the impact of prostaglandin use and differences in corneal biomechanics between eyes with high-tension versus low-tension glaucoma.
Although the current approach to treating glaucoma centers on reducing IOP, many patients with ocular hypertension never develop glaucoma. Conversely, up to one-third of patients with glaucoma have baseline IOPs below 21 mm Hg, in the so-called normal range. This is likely due to individual differences in corneal and scleral biomechanical properties. It is not the applied stress of IOP that causes glaucomatous damage but rather the subsequent optic nerve deformational changes or strain. Scleral biomechanical properties are difficult to assess owing to location and the overlying conjunctiva. Corneal biomechanical properties are easier to evaluate and may aid in risk stratification.
Two devices are currently used worldwide for the assessment of corneal biomechanics in vivo: the Ocular Response Analyzer (ORA, Reichert Technologies) and the Corneal Visualization Scheimpflug Technology (Corvis ST, Oculus).1,2 They report different, albeit related, corneal biomechanical parameters.
In brief, the ORA evaluates the bidirectional corneal movement in response to an air jet. The main parameter reported by the device is corneal hysteresis (CH), a measure of the cornea’s ability to dissipate energy (ie, dampening corneal capacity). Other reported parameters are corneal resistance factor (CRF), Goldmann-corrected IOP, and corneal-compensated IOP (IOPcc; Figure 1).3 CH may be affected by various factors, including central corneal thickness, age, diabetes, axial length, keratoconus, and, more important, IOP; these factors may confound results and are not accounted for in many studies of the ORA.4
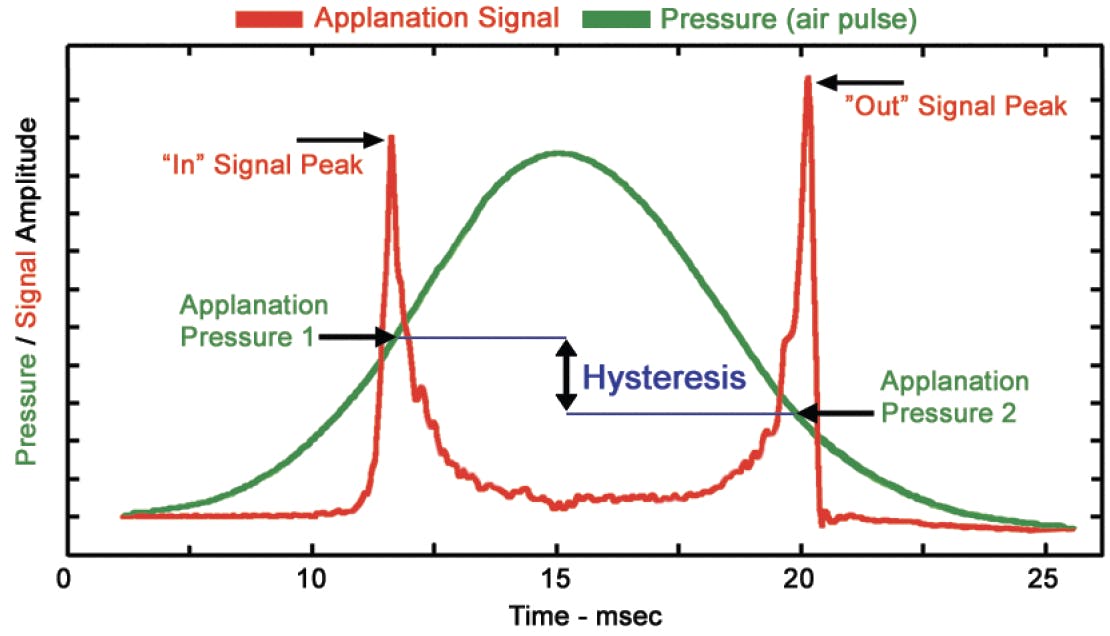
Figure 1. The applanation signal and air pulse pressure diagram obtained over the course of one measurement. Applanation pressure 1 is the pressure at which the cornea reaches a specific applanation state on inward movement. Applanation pressure 2 is the pressure at which the cornea passes through this applanation state on outward movement. The difference between the two is the CH parameter, which is the main output of the ORA.
(Courtesy of Reichert Technologies)
The Corvis ST evaluates corneal biomechanics by imaging with a Scheimpflug camera corneal deformational changes in response to an air jet (Figure 2). The device evaluates more than 35 parameters, which, for simplicity, can be classified into two groups. First are those affected mainly by IOP rather than corneal stiffness; these include applanation time, velocity, and deformational amplitude (DA). The second group is affected mainly by corneal stiffness rather than IOP. Examples include DA ratio, stress-strain index, integrated inverse radius, stiffness parameter at first applanation, and stiffness parameter at highest concavity.5

Figure 2. The deformation process captured by the Corvis ST. The recording starts with the cornea in a natural convex shape (A). A precisely metered air pulse forces the cornea to move inward through an applanation (applanation 1; B). The cornea continues to move inward until it reaches the greatest concavity (C). Because of its viscoelasticity, the cornea rebounds from its greatest concavity to its normal convex curvature and again passes through an applanation (applanation 2; D). Reprinted with permission from Tian et al.18
In primary open-angle glaucoma and healthy controls, the correlation between the ORA-measured CH and Corvis ST–measured parameters has been reported to be weak to moderate.6
CORNEAL BIOMECHANICS IN GLAUCOMATOUS EYES
Several studies have reported that CH is significantly lower in patients with glaucoma compared to healthy controls. This suggests that CH measurements may be of value in clinical practice. Eyes with low CH have a diminished ability to dissipate energy in response to applied stress (ie, increased IOP or IOP fluctuations) and are presumably more susceptible to glaucomatous damage. However, CH is significantly associated with IOP, so comparing glaucomatous eyes with high IOPs to control eyes with lower baseline IOPs, for example, may confound study interpretation. Based on Laplace’s law, the higher the IOP, the stiffer and “less deformable” the cornea. In other words, in the presence of ocular hypertension, an originally softer cornea behaves more stiffly than an originally stiffer cornea at a lower IOP. Studies designed to account for the effect of IOP on corneal biomechanics could help establish corneal biomechanics as a clinical tool.
Most research on CH in glaucoma to date has included patients who were administering topical prostaglandin analogues. These agents may induce structural changes in the corneal extracellular matrix structure, resulting in lower CH and ultimately confounding any significant association between CH and glaucoma.4,5 Studies have shown that evaluating CH can help predict a glaucoma suspect’s risk of developing glaucoma and the rate of visual field progression in a patient with the disease. In a prospective longitudinal study, 19% of 287 glaucoma suspect eyes developed glaucomatous visual field defects over a mean follow-up period of almost 4 years. The study authors showed that CH was significantly lower in eyes that developed glaucoma than in those that did not (9.5 vs 10.2 mm Hg). After adjusting for confounding factors including IOP, CH was a significant predictor of glaucoma development (hazard ratio, 1.2).7 This study suggests that low CH is a significant risk factor for glaucoma development.
Measuring baseline CH and tracking CRF over time may provide important information about an individual’s risk of glaucomatous progression. Patients with glaucoma and low baseline CH are at increased risk of faster visual field progression. One study of patients with glaucoma demonstrated that each 1 mm Hg decrease in CH was associated with a 0.25%/y faster rate of visual field index decline over time.8 Another study showed that the CRF reduction over time was significantly associated with faster visual field progression.9 Lower CH and CRF may be associated with reduced peripapillary scleral stiffness, making the optic nerve more susceptible to damage from increased IOP with subsequent visual field progression.10
Another important factor measured by the ORA is IOPcc, which compensates for corneal artifacts in an attempt to provide more accurate values than IOP measured by Goldmann applanation tonometry. A prospective study found IOPcc to have a stronger association with visual field progression in glaucomatous eyes than IOP measured by Goldmann applanation tonometry or iCare rebound tonometry (Icare USA).11 Other studies have reported an association between low CH and optic disc hemorrhage12 and disc surface depression, which has been hypothesized to occur before notable thinning of the retinal nerve fiber layer.13
Some studies have found that the corneas of glaucomatous eyes are more deformable than those of healthy controls.14,15 Other studies have reported the opposite.16,17 Pradhan et al showed that, after adjusting for IOP, there was no significant difference in the Corvis ST parameters among eyes with primary open-angle glaucoma, eyes with pseudoexfoliation glaucoma, and healthy controls.19 Some differences in these reports may be attributable to the evolution of the parameters and the software used to measure corneal stiffness; the latest software uses parameters that are less affected by IOP.
In glaucoma suspects, the higher the stiffness parameter at first applanation or the stiffer the cornea, the faster the rate of thinning of the retinal nerve fiber layer and ganglion cell–inner plexiform layer.20 Published research on the association between Corvis ST–measured parameters and visual field progression in glaucomatous eyes is limited, and results are conflicting.21,22 One study reported no significant association between Corvis ST parameters and visual field progression, whereas another demonstrated an association between a higher DA ratio and a worse visual field.
CONCLUSION
The assessment of corneal biomechanics may come to play an important role in glaucoma management and risk stratification. Current methods of assessing corneal biomechanics are promising, but they are limited to some degree by the confounding effects of IOP. Future studies, moreover, must take into account the impact of prostaglandin use and differences in corneal biomechanics between eyes with high-tension versus low-tension glaucoma.
1. Jammal AA, Medeiros FA. Corneal hysteresis: ready for prime time? Curr Opin Ophthalmol. 2022;33:243-249.
2. Lopes BT, Elsheikh A. In vivo corneal stiffness mapping by the stress-strain index maps and Brillouin microscopy. Curr Eye Res. 2022;1-7.
3. Roberts CJ. Concepts and misconceptions in corneal biomechanics. J Cataract Refract Surg. 2014;40:862-869.
4. Sit AJ, Chen TC, Takusagawa HL, et al. Corneal hysteresis for the diagnosis of glaucoma and assessment of progression risk: a report by the American Academy of Ophthalmology. Ophthalmology. Published online December 16, 2022. doi:10.1016/j.ophtha.2022.11.009
5. Shen SR, Fleming GP, Jain SG, Roberts CJ. A review of corneal biomechanics and scleral stiffness in topical prostaglandin analog therapy for glaucoma. Curr Eye Res. 2022;1-10.
6. Fujishiro T, Matsuura M, Fujino Y, et al. The relationship between Corvis ST tonometry parameters and Ocular Response Analyzer corneal hysteresis. J Glaucoma. 2020;29:479-484.
7. Susanna CN, Diniz-Filho A, Daga FB, et al. A prospective longitudinal study to investigate corneal hysteresis as a risk factor for predicting development of glaucoma. Am J Ophthalmol. 2018;187:148-152.
8. Medeiros FA, Meira-Freitas D, Lisboa R, Kuang TM, Zangwill LM, Weinreb RN. Corneal hysteresis as a risk factor for glaucoma progression: a prospective longitudinal study. Ophthalmology. 2013;120;1533-1540.
9. Chan E, Yeh K, Moghimi S, et al. Changes in corneal biomechanics and glaucomatous visual field loss. J Glaucoma. 2021;30:e246-e251.
10. Girard MJ, Suh JK, Bottlang M, Burgoyne CF, Downs JC. Biomechanical changes in the sclera of monkey eyes exposed to chronic IOP elevations. Invest Ophthalmol Vis Sci. 2011;52:5656-5669.
11. Susanna BN, Ogata NG, Daga FB, Susanna CN, Diniz-Filho A, Medeiros FA. Association between rates of visual field progression and intraocular pressure measurements obtained by different tonometers. Ophthalmology. 2019;126:49-54.
12. Radcliffe NM, Tracer N, De Moraes CGV, Tello C, Liebmann JM, Ritch R. Relationship between optic disc hemorrhage and corneal hysteresis. Can J Ophthalmol. 2020;55:239-244.
13. Xu G, Chen Z. Corneal hysteresis as a risk factor for optic nerve head surface depression and retinal nerve fiber layer thinning in glaucoma patients. Sci Rep. 2021;11:11677.
14. Miki A, Yasukura Y, Weinreb RN, et al. Dynamic Scheimpflug ocular biomechanical parameters in healthy and medically controlled glaucoma eyes. J Glaucoma. 2019;28:588-592.
15. Hong K, Wong IYH, Singh K, Chang RT. Corneal biomechanics using a Scheimpflug-based noncontact device in normal-tension glaucoma and healthy controls. Asia Pac J Ophthalmol (Phila). 2019;8:22-29.
16. Coste V, Schweitzer C, Paya C, Touboul D, Korobelnik JF. Evaluation of corneal biomechanical properties in glaucoma and control patients by dynamic Scheimpflug corneal imaging technology. Article in French. J Fr Ophtalmol. 2015;38:504-513.
17. Wang W, Du S, Zhang X. Corneal deformation response in patients with primary open-angle glaucoma and in healthy subjects analyzed by Corvis ST. Invest Ophthalmol Vios Sci. 2015;56:5557-5565.
18. Tian L, Wang D, Wu Y, et al. Corneal biomechanical characteristics measured by the Corvis Scheimpflug technology in eyes with primary open-angle glaucoma and normal eyes. Acta Ophthalmologica. 2016;94:e317-e324.
19. Pradhan ZS, Deshmukh S, Dixit S, et al. A comparison of the corneal biomechanics in pseudoexfoliation glaucoma, primary open-angle glaucoma and healthy controls using Corvis ST. PLoS One. 2020;15:e0241296.
20. Qassim A, Mullany S, Abedi F, et al. Corneal stiffness parameters are predictive of structural and functional progression in glaucoma suspect eyes. Ophthalmology. 2021;128:993-1004.
21. Bolivar G, Sanchez-Barahona C, Ketabi S, Kozobolis V, Teus MA. Corneal factors associated with the amount of visual field damage in eyes with newly diagnosed, untreated, open-angle glaucoma. Ophthalmol Ther. 2021;10:669-676.
22. Vinciguerra R, Rehman S, Vallabh NA, et al. Corneal biomechanics and biomechanically corrected intraocular pressure in primary open-angle glaucoma, ocular hypertension and controls. Br J Ophthalmol. 2020;104:121-126.




