CASE PRESENTATION
A 57-year-old White woman had chronic nongranulomatous panuveitis and cystoid macular edema (CME) in her right eye despite treatment with systemic methotrexate and topical prednisolone and nepafenac. The patient had been diagnosed with rheumatoid arthritis and tested positive for HLA-B27. She developed a steroid response, and her IOP rose to 45 mm Hg OD on four classes of topical antiglaucoma medication and acetazolamide 250 mg dosed four times per day.
A nuclear and posterior subcapsular cataract was present in the patient’s right eye, and her UCVA was 20/200 OD. Testing with a potential acuity meter suggested that her UCVA might improve to 20/60 after cataract extraction.
On examination, the angles were narrow with only anterior trabecular meshwork visible over 270º. The anterior chamber of the right eye was quiet on the current regimen of prednisolone 1% dosed three times daily, nepafenac, and methotrexate. The view to the posterior segment of the eye was poor through the cataract. The cup-to-disc ratio was estimated to be 0.6, and no frank CME was evident. Visual field progression had occurred during the recent IOP rise, and advanced loss splitting fixation was observed (Figure 1).
To foster more robust discussion, we are breaking with the column’s usual format this issue and allowing panelists to read in advance how the case was managed and see its outcome. Details on the management of this case therefore directly follow the presentation.
—Case prepared by Devesh K. Varma, MD, FRCSC
WHAT I DID: DEVESH K. VARMA MD, FRCSC
A patient with chronic uveitis, a strong steroid response, and rapid visual field progression would give any glaucoma surgeon pause, particularly if the patient were as young as this one. I counseled her that surgery was required and that her prognosis was guarded.
The potential acuity measurement demonstrated room for improvement, and the IOP was uncontrolled on maximum tolerated medical therapy. I therefore recommended a cataract procedure combined with glaucoma filtering surgery. The anterior chamber was quiet, and there was no CME; the timing for surgery was therefore reasonable. Because of the patient’s history of uveitis and CME, I was concerned she might have a volatile postoperative course complicated by CME, hypotony, or fibrosis. I thought that an Ahmed Glaucoma Valve (New World Medical) or PreserFlo MicroShunt (Santen) might provide more predictable outflow than a trabeculectomy. I opted for the PreserFlo owing to its bioinert poly(styrene-block-isobutylene-block-styrene) or SIBS material, which I hoped would reduce the risk of fibrosis. I also hoped the implant’s thin profile would minimize the chance of future erosion and exposure.
Cataract surgery was performed, and a PreserFlo MicroShunt was implanted after a 2-minute application of 0.4 mg/mL mitomycin C. Postoperatively, the patient administered topical dexamethasone every 2 hours, which was tapered over 6 weeks. One week after surgery, the IOP was 8 mm Hg on no glaucoma drops and remained at a similar level thereafter. Two months after surgery, the patient’s UCVA was 20/30+2 OD, and the visual field for that eye showed marked improvement (Figure 2). Despite evidence of significant retinal nerve fiber layer (RNFL) loss on OCT imaging (Figure 3), I reclassified the severity of the patient’s glaucoma as moderate based on the visual field. At her last visit, 6 months after surgery, the IOP was 9 mm Hg off glaucoma drops.
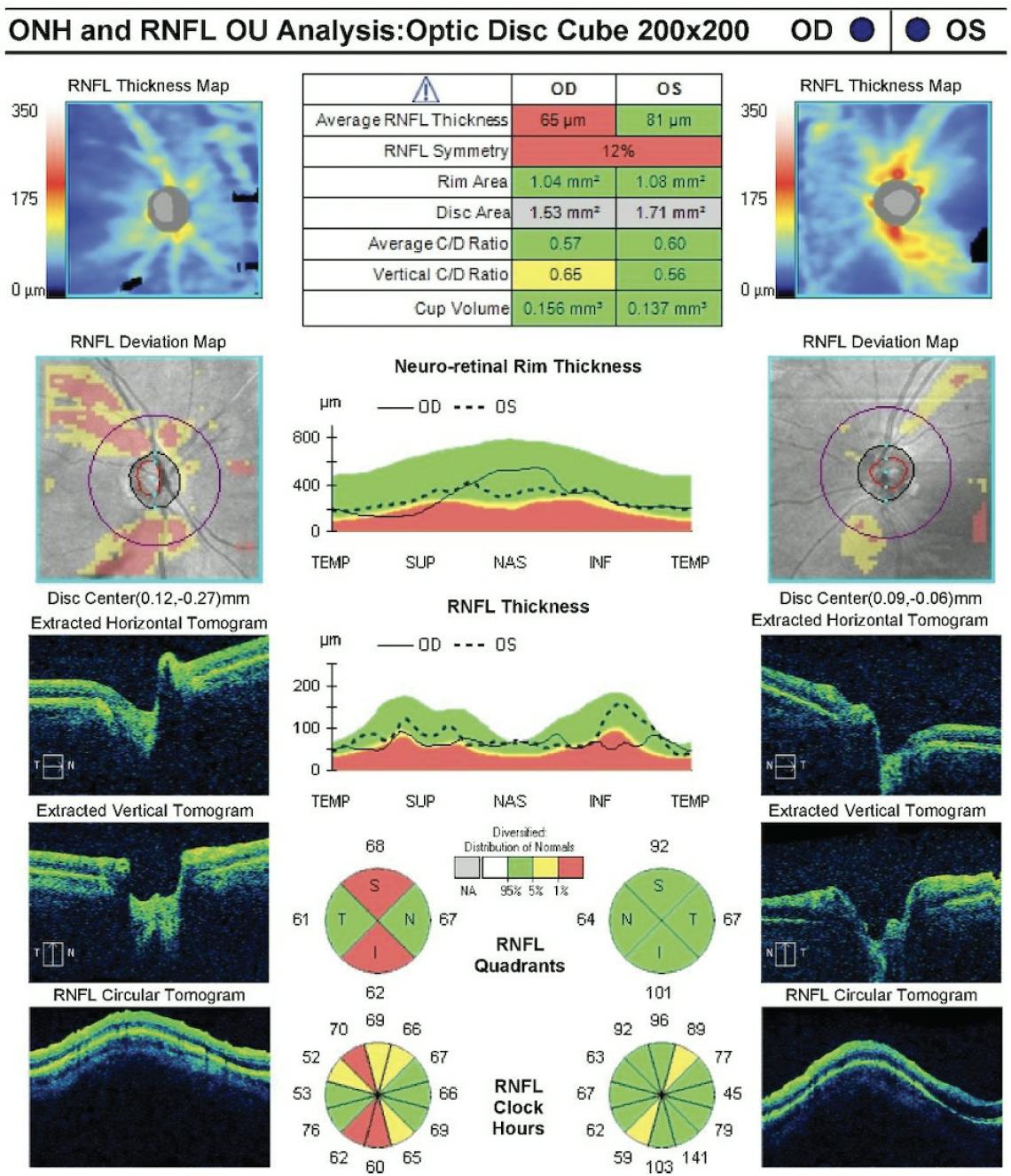
Figure 3. OCT imaging obtained 2 months after surgery showed marked superior and inferior RNFL loss in the right eye.
RONALD L. FELLMAN, MD
Uncontrolled uveitic glaucoma accompanied by a significant cataract requires high-level surgical decision-making. Fortunately, before surgery, the patient’s inflammation seemed to be under reasonable control with topical steroids and immunomodulatory therapy. The angle was apparently open but might have been prone to the development of postoperative peripheral anterior synechiae.
Canal-based angle surgery is a reasonable option for steroid-induced glaucoma. An overall diagnosis of panuveitis and an inability to see the trabecular area well, however, can reduce the efficacy of angle surgery.
Cataract extraction alone in a uveitic eye may worsen the inflammation and cause a postoperative IOP spike, but this can be mitigated by the placement of a filter or drainage device. The PreserFlo MicroShunt that Dr. Varma selected was an excellent choice in this case, but it is not available in the United States, where I practice. A Xen45 Gel Stent (Allergan) has a diameter of 45 µm and may become blocked by fibrin related to uveitis, so this device would not have been the best option here. Standard trabeculectomy has a poor long-term success rate in uveitic eyes. A good alternative would have been an Ahmed Glaucoma Valve FP7. Its valved design allows an immediate IOP reduction, and its surface area is compatible with preventing hypotony that may be related to a long-term reduction in aqueous production that can occur with uveitis. An intraoperative sub-Tenon injection of a long-lasting steroid may help to prevent retinal edema.
The patient’s visual field improvement is exciting and likely the result of improved IOP control in conjunction with cataract extraction. Visual field improvement is highly variable after combined cataract and glaucoma surgery. The greatest improvement occurs when both surgeries are successful and the optic nerve has not been severely damaged.
ANDREW GROSS, MD, AND KELLY W. MUIR, MD, MHSC
Concurrent cataract and uveitic glaucoma are usually challenging. In this case, preoperative visual field testing (Figure 1) indicates bihemispheric depression. A clinical examination of the optic nerve and OCT findings (Figure 3) suggests that not all the visual loss was attributable to glaucoma. The cataract was perhaps visually significant, likely to worsen, and contributing to narrow angles.
One option was to perform cataract extraction alone, but with the preoperative IOP level and presumed need for future steroid therapy, our preference would have been to perform concurrent cataract and filtering surgery. Given the patient’s young age and quiet eye, trabeculectomy or the placement of a Xen Gel Stent was a reasonable strategy, but the risk of long-term scarring would have inclined us toward a glaucoma drainage device. For uveitic eyes, we generally favor a valved implant because it may carry less risk of hypotony than an unvalved implant.
Our discussion with the patient would have addressed two main points: (1) visual outcome and (2) IOP control. Uveitic glaucoma is difficult to stage because visual fields can be obfuscated by cataracts and CME. Furthermore, edema of the RNFL may hide glaucomatous OCT changes or make optic nerve cups seem smaller on clinical examination.2 Pertinent to the discussion of future IOP control, failure requiring additional surgery or more aggressive topical medical therapy is a common scenario.3
We suspect that the visual field improvement the patient experienced after surgery was due in greater part to cataract removal than to a reversal of glaucomatous field loss, but improvement is always welcome nonetheless.








