A Cystotome Where It Should Not Be
By Deidre St. Peter, MD, and Cara E. Capitena Young, MD
A 75-year-old patient was scheduled for phacoemulsification in the right eye at a tertiary hospital. The surgeon (a new trainee) had started to perform the capsulorhexis when they suddenly lost control of the cystotome while attempting to rotate the instrument. The cystotome shot forcibly across the anterior chamber and around the equator of the lens (Figure 1).
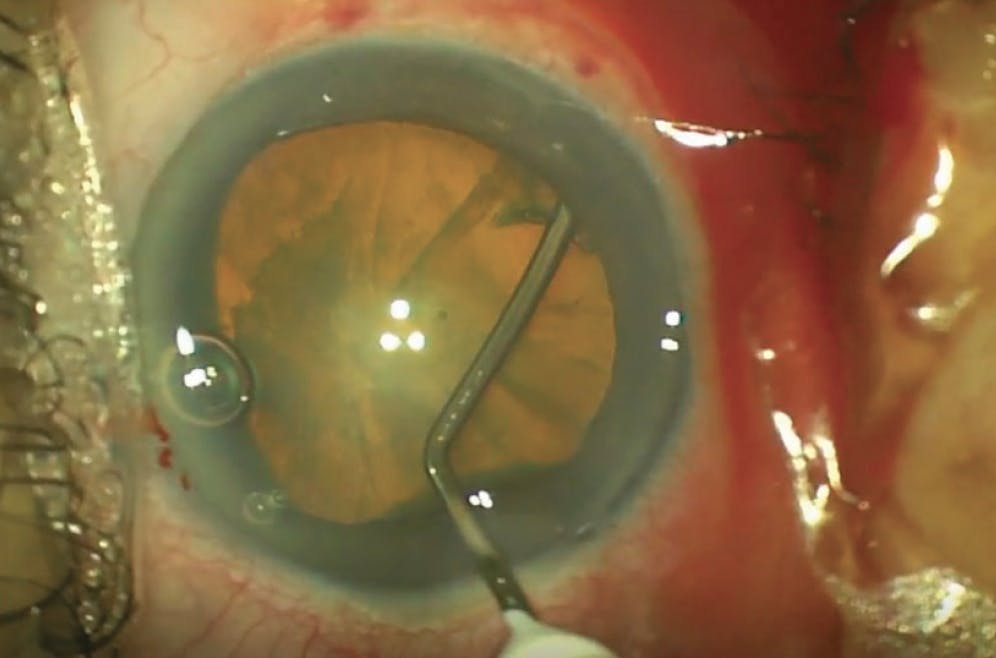
Figure 1. A surgical trainee lost control of a cystotome, causing the instrument to shoot forcibly across the anterior chamber and around the lens equator.
Upon retraction of the cystotome, a strand of vitreous was identified at the instrument’s edge. Intraocular scissors were used to lyse the vitreous strand and remove the cystotome. Triamcinolone acetonide (Kenalog, Bristol-Myers Squibb) was injected, and no further vitreous was identified. The capsule appeared to be intact (see Watch Now). The capsulorhexis was completed carefully, and phacoemulsification was performed without issue, although caution was taken to prevent fluctuations in anterior chamber depth by using additional OVD tamponade in the area of the incident. Manipulation of lens pieces in that area was avoided when possible.
After cataract removal, a one-piece IOL was placed in the bag, and the haptics were oriented away from the inferonasal sector. Triamcinolone was again injected, and no vitreous was identified in the anterior chamber. Triamcinolone traveled posteriorly into the vitreous, confirming a focal area of zonular loss at the site of cystotome impact. The patient did well postoperatively, with a small strand of vitreous noted in the sulcus but none observed anteriorly (Figure 2).

Figure 2. Extra care was taken to complete the capsulorhexis and perform phacoemulsification. A one-piece IOL was placed in the bag with the haptics oriented away from the inferonasal sector. Postoperatively, a small strand of vitreous was noted in the sulcus, but none was observed anteriorly.
In unforeseen circumstances, knowledge of how to handle vitreous among other complications properly—and a bit of intraoperative patience—can lead to successful visual outcomes.
Hypotony After Nonvalved Tube Shunt Surgery
By Timothy Truong, MD, and Craig J. Chaya, MD
A 13-year-old patient presented for a 6-week postoperative visit with hypotony and choroidal effusion from overfiltration after nonvalved tube shunt surgery.
An earlier firework injury had left the eye with a dislocated crystalline lens, lens-cornea touch, iridodialysis, traumatic mydriasis, and a hyphema. The globe was found to be intact. The patient had undergone an emergency lensectomy and anterior vitrectomy, and the eye was left aphakic.
Although the patient initially did well after emergency surgery, he returned for a postoperative week 1 appointment with an eight-ball hyphema, vitreous hemorrhage, and an IOP of 38 mm Hg despite maximum medical therapy. He then underwent a full pars plana vitrectomy and placement of a nonvalved tube shunt in the pars plana. A large submacular hemorrhage was noted intraoperatively.
The first month’s postoperative course was uneventful. At week 6, however, the patient presented to his local ophthalmologist with a flat chamber, hypotony, and choroidal effusion, and he was emergently referred to us for surgical intervention.
The patient was placed under general anesthesia. A robust, diffuse superotemporal bleb was noted, and it was suspected that the nonvalved tube shunt had fully opened without significant scar tissue formation, resulting in overfiltration and hypotony. An OVD injection with a 30-gauge needle was performed to stabilize the eye before multiple paracentesis incisions were made. An anterior chamber maintainer was placed, and the IOP was set to 30 mm Hg to help reduce the choroidal effusion.
Three approaches were undertaken to remedy the situation (see Watch Now). We first attempted the use of an ab interno polypropylene suture plug to reduce aqueous flow. A 3-0 polypropylene suture (Prolene, Ethicon) was threaded into the tube shunt device. Microforceps were used to mark the polypropylene suture at the appropriate length. The suture was then externalized, cut to size, and flanged with low-temperature cautery. Microforceps and micrograspers were used to rethread the polypropylene plug into the tube. Despite these steps, we determined that the bleb remained robust, and the eye was at high risk for continued hypotony. The polypropylene plug was therefore removed from the eye.
We then proceeded with an ab interno attenuation of the tube. A paracentesis incision was made on the cornea anterior to the location of the tube, and the distal tip of the device was externalized through the paracentesis. A 6-0 polypropylene suture was used to attenuate the tube, which was then challenged successfully with balanced salt solution on a 27-gauge cannula to confirm restricted flow before replacement in the eye. Although flow was significantly reduced, the tube’s position was too anterior due to the knot’s location. The tube was then brought back through the paracentesis, and the suture was cut.
Our final solution was to reopen the prior envelope conjunctival incision to attenuate the tube shunt in an ab externo fashion. A cutdown of conjunctiva and Tenon capsule was undertaken until a gush of fluid was noted, confirming limited scar tissue formation despite a 6-week healing period. The corneal patch graft was elevated carefully to expose the tube shunt, which was found to be in good condition. The tube was tied off with a 6-0 polypropylene suture, and minimal flow was noted. The corneal patch graft was resutured to the sclera, and the conjunctival-Tenon incision was closed with 10-0 polyglactin sutures (Vicryl, Ethicon). All corneal incisions were also closed with 10-0 polyglactin sutures.
The choroidal effusions had resolved almost completely by the postoperative day 1 visit. The patient’s IOP was stable at 13 mm Hg at the most recent visit, 6 months after surgery, on one drop of a fixed combination of dorzolamide and timolol administered twice daily. The choroidal effusions had resolved completely. His visual acuity remained counting fingers, limited by the submacular hemorrhage and scarring.
This case involved a young patient, but we have used these techniques successfully in both pediatric and adult eyes.
Alternating Quadrants for Stent Implantation
By Arkadiy Yadgarov, MD
I recently had a case in which a stricture in Schlemm canal precluded the superonasal implantation of a Hydrus Microstent (Alcon). As shown in the video (see Watch Now), the cannula was being pushed back because the stent could not propagate forward. I removed the cannula from the eye and turned it in the opposite direction to attempt inferonasal implantation instead. The Hydrus was able to cannulate with much more ease in this quadrant. Anatomic variations in Schlemm canal may sometimes necessitate a change in surgical plans.
Three Techniques for Guidewire-Assisted Tube Shunt Placement in the Ciliary Sulcus
By Jessie Wang, MD; Arjav Shah, MD; Jacob A. Kanter, MD; Jonathan Eisengart, MD; Lauren S. Blieden, MD; and Mary Qiu, MD
Tube shunts can be inserted into the anterior chamber, pars plana, or ciliary sulcus. Compared to posterior placement, anterior chamber placement carries a theoretical risk of corneal decompensation, whereas insertion in the pars plana requires concurrent or preexisting vitrectomy.1 The ciliary sulcus may therefore represent a desirable middle ground for tube shunt insertion.2
The sulcus-based approach is generally reserved for pseudophakic eyes, given the narrow space between the crystalline lens and the posterior iris. Even in pseudophakic eyes, sulcus placement may be challenging. If too anterior, the needle may enter the anterior chamber or cause an iridodialysis. If too posterior, the needle may dislodge the IOL or enter the vitreous cavity. Sometimes the rigid needle enters the sulcus, but the soft tube goes behind the IOL. Given these challenges, we describe three techniques for guidewire-assisted tube shunt placement in the ciliary sulcus (Figure) and showcase them in the accompanying video (see Watch Now).3

Figure. Three techniques for guidewire-assisted tube shunt insertion in the ciliary sulcus, as demonstrated by Dr. Eisengart (A), Dr. Blieden (B), and Dr. Qiu (C).
Technique No. 1
A 23-gauge needle is inserted through a paracentesis 180° across from the planned tube entry site and used to make an ab interno sclerotomy. A guidewire is threaded into the needle bevel, and the needle is retracted to pull the guidewire into the sulcus. This approach eliminates blind needle entry into the sulcus space, thereby reducing the risk of damage to the iris root, zonular fibers, and capsular bag.
Technique No. 2
A paracentesis is made at any convenient location, and a guidewire is inserted into the eye. A 23-gauge needle is used to make a sclerotomy into the sulcus. Microforceps are inserted into the sclerotomy to retrieve the guidewire and pull it out of the eye. This technique can be especially useful in eyes in which it is not anatomically feasible to make a paracentesis 180° away from the planned tube entry site (eg, those with a history of corneal surgery).
Technique No. 3
A paracentesis is made 180° away from the planned tube entry site, and a guidewire is inserted through the paracentesis into the anterior chamber. A 23-gauge needle is used to make a sclerotomy into the sulcus. The guidewire is grasped with tying forceps outside of the eye with the surgeon’s nondominant hand and docked into the bevel of the needle inside the eye, which is held by the surgeon’s dominant hand. The needle is retracted to pull the guidewire out of the eye. Like the second technique, this one allows the sclerotomy to be made using the familiar ab externo approach.
With all three techniques, the tube is then threaded onto the guidewire and inserted into the sclerotomy. The guidewire is then removed from the paracentesis, leaving the sulcus tube in place. We hope that sharing these varying approaches helps to facilitate consistent tube shunt insertion in the ciliary sulcus.
1. Zhang Q, Liu Y, Thanapaisal S, et al. The effect of tube location on corneal endothelial cells in patients with Ahmed Glaucoma Valve. Ophthalmology. 2021;128(2):218-226.
2. Tello C, Espana EM, Mora R, Dorairaj S, Liebmann JM, Ritch R. Baerveldt Glaucoma Implant insertion in the posterior chamber sulcus. Br J Ophthalmol. 2007;91(6):739-742.
3. Shah A, Kanter JA, Eisengart J, Blieden LS, Qiu M. Three techniques for guidewire-assisted sulcus glaucoma tube shunt placement. Am J Ophthalmol Case Rep. 2024;34:102009.
Hemorrhage From Iris Root Trauma During GATT
By Sirjhun Patel, MBChB, BSc, FRCOphth, and Irfan N. Kherani, AB, MD, FRCSC
A 74-year-old man was being treated for advanced primary open-angle glaucoma in his left eye. His preoperative IOP was 24 mm Hg OS on four classes of medications. The patient underwent cataract surgery combined with a 180° gonioscopy-assisted transluminal trabeculotomy (GATT) in the left eye.
The video shows the GATT portion of the surgery (see Watch Now). A microvitreoretinal blade was used to perform a goniotomy. A 5-0 polypropylene suture, with its tip cauterized, was held with micrograspers and introduced into the goniotomy site. However, during this process, the micrograspers inadvertently moved posteriorly and came in contact with the iris, resulting in traumatic bleeding from the iris root.
To manage the hemorrhage, a cohesive OVD was strategically injected inferiorly to displace the blood superiorly (moving it from left to right). Care was taken to prevent the blood from dispersing, which could have further obscured the view of the angle. The OVD also temporarily increased the IOP, tamponading the bleeding and preventing additional blood from entering the anterior chamber.
Once hemostasis was achieved and a clear view of the angle was restored, the polypropylene suture was successfully advanced into Schlemm canal. It was then fed 180° around the canal using the same micrograspers. After moving the patient’s head into a neutral position, the polypropylene suture was withdrawn from the eye, completing a 180° goniotomy.
The OVD and residual blood were removed using automated irrigation and aspiration. The eye was kept pressurized with balanced salt solution on a 27-gauge cannula through the sideport incision during removal of the I/A probe, and the eye was left hyperpressurized to prevent further blood reflux into the angle.
JOIN THE CONVERSATION
Surgical complexity of your own to share? Submit it to Eyetube.
https://eyetube.net/video/submit




















