CASE PRESENTATION
A 49-year-old man presents for a routine glaucoma follow-up visit. He has a history of birdshot chorioretinopathy and related secondary glaucoma.
The patient underwent bilateral cataract surgery 10 years ago. One year ago, the IOP in both eyes rose into the low 30s mm Hg despite maximal medical therapy, and he underwent a 180º goniotomy bilaterally. Following surgery, the left eye maintained an IOP in the midteens. The right eye, however, experienced an initial IOP spike into the 40s mm Hg. Maximal drop therapy with acetazolamide, a fixed combination of brimonidine and timolol, netarsudil, and latanoprost ophthalmic solution was required before the IOP settled into the low teens. The patient has received two fluocinolone acetonide intravitreal implants 0.18 mg (Yutiq, Alimera Sciences) in the right eye and one in the left eye to treat his birdshot chorioretinopathy. Each eye received one implant 3 years ago, and the second implant in the right eye was placed 1 month ago.
His current drug regimen consists of a fixed combination of brinzolamide and brimonidine tartrate ophthalmic suspension, netarsudil, and latanoprost ophthalmic solution in both eyes and timolol administered once daily in the morning in the right eye only.
Upon presentation, the patient’s BCVA is 20/20 OU. His IOP is 23 mm Hg OD and 12 mm Hg OS. An examination of each eye finds a deep, quiet anterior chamber; a well-centered posterior chamber IOL in the capsular bag; and slight pseudophacodonesis. Gonioscopy shows grade 4 angles with a yellow cylindrical foreign body in the inferior angle of the right eye (Figure 1). A fundus examination finds optic nerve cupping bilaterally that is greater in the right eye and classic findings of birdshot chorioretinopathy (Figure 2).
Humphrey visual field testing (Carl Zeiss Meditec) shows a constricted visual field in both eyes (Figure 3). OCT imaging reveals severe cupping and thinning of the retinal nerve fiber layer in both eyes (Figure 4).
The questions posed to the panelists depended on whether they are glaucoma or uveitis specialists. Drs. Aref and Arosemena were asked how they would improve IOP control in the right eye and address the steroid implant that has migrated into the anterior chamber. Dr. Patel was asked what he would advise regarding the implants in the anterior chamber and managing the patient’s uveitis in the future.
—Case prepared by Jacob Brubaker, MD
AHMAD A. AREF, MD, MBA
The uncontrolled glaucoma in the patient’s right eye appears to be related to his recent intravitreal steroid therapy. The IOP in the left eye, which did not receive a steroid implant recently, has been controlled with topical medications since undergoing angle-based surgery.
Glaucoma filtering surgery is indicated in the patient’s right eye given the advanced nature of his disease, history of angle-based surgery, and treatment with multiple topical agents. My preference would be to place a nonvalved aqueous shunt, either an Ahmed Clearpath Implant (New World Medical) or a Baerveldt Glaucoma Implant (Johnson & Johnson Vision). A nonvalved device is more likely to be successful in this situation considering the patient’s age and his increased risk of episcleral fibrosis, failure with trabeculectomy, hypermature capsule formation after the implantation of a Xen Gel Stent (AbbVie), and failure with implantation of a valved aqueous shunt such as an Ahmed Glaucoma Valve (New World Medical).
My role as a glaucoma consultant is to manage the IOP while continuing the indicated steroid therapy. The steroid implants may be left in the anterior chamber if a uveitis specialist sees no clinical evidence of corneal decompensation or indication for removal.
ANALISA AROSEMENA, MD
Balancing the risk and benefits of uveitis and glaucoma treatment can be challenging. Controlling uveitic inflammation comes first. Although immunomodulators are available, steroids are often required, worsening IOP control.1
The success of uveitis control with extended-release steroids is high.1,2 IOP elevation, however, has been reported in 50% to 100% of patients receiving this form of treatment, and in a few studies, more than 36.6% of patients required glaucoma surgery at 3 years.1-3 Although Yutiq is better tolerated than other biodegradable steroid implants, it must be removed if it migrates into the anterior chamber.4
The patient has severe visual field loss from glaucoma and birdshot chorioretinopathy. The IOP in the right eye must be controlled. Selective laser trabeculoplasty can be an effective treatment for steroid-induced glaucoma,5 but the procedure has minimal efficacy after a 180º goniotomy.6 The fluocinolone acetonide intravitreal implant would therefore be removed from the anterior chamber with microforceps via a large paracentesis. A valved drainage device such as an Ahmed Glaucoma Valve would then be implanted to prevent hypotony.
It might be necessary for a retina specialist to place another steroid implant in the right eye, but the risk of migration should be reassessed because the pseudophacodonesis will still be present.
SARJU PATEL, MD, MPH, MSC
This fairly young patient has severe, end-stage birdshot chorioretinopathy. Although he has secondary glaucoma, the cupping, pallor, and field loss are partially due to chorioretinopathy with secondary atrophy (as evidenced by the dense temporal field loss in the left eye).
I would educate the patient on birdshot chorioretinopathy and the high potential for severe vision loss and implore him to start systemic treatment. With disease this severe, local steroid therapy, while useful, should be used as an adjunct if immunotherapy cannot achieve full control. The exception would be if a fluocinolone acetonide intravitreal implant 0.59 mg (Retisert, Bausch + Lomb) were placed. Even then, however, the patient would likely need additional immunotherapy. Moreover, given the IOP issues, the option of local steroids would be less attractive. That said, if steroids are required, I would stay away from intravitreal implants. The risk of repeat anterior chamber migration is high, and the patient’s right eye would not do well with additional corneal surgery if the endothelium is lost. Although this occurs less often with Yutiq compared to a dexamethasone intravitreal implant 0.7 mg (Ozurdex, AbbVie), the risk is unacceptable when other excellent options such as triamcinolone acetonide suprachoroidal injectable suspension (Xipere, Bausch + Lomb), a sub-Tenon injection of triamcinolone, and oral corticosteroids are available.
As for immediate management, the pupil of the right eye would be dilated, and the patient’s face would be manipulated until the implant migrates posteriorly. If repositioning is successful, the patient’s sleeping habits and other activities would be reviewed to restrict face-down positioning. If the implant migrates again, then surgical removal would be required.
WHAT I DID: JACOB BRUBAKER, MD
This case speaks to the challenge of managing uveitic glaucoma. A fluocinolone acetonide intravitreal implant had migrated through the zonules and into the anterior chamber. I felt the necessary first step was to remove the implant to prevent damage to the corneal endothelium. I also postulated that the implant’s proximity to the trabecular meshwork might be increasing the steroid response. Upon inspecting the angles, I hypothesized that either the earlier 180º goniotomy treatment had been incomplete or the trabecular meshwork had healed over in many locations.
The patient and I discussed two options: (1) removing the fluocinolone acetonide intravitreal implant and enhancing the goniotomy and (2) combining implant removal with the placement of a glaucoma drainage device. Repeat goniotomy has been shown to be successful in a pediatric population.7 Although the patient was an adult, I felt that, given the minimal IOP elevation and the gonioscopic appearance of the eye, an enhancement would be a reasonable first step. He elected to undergo repeat goniotomy at the time of implant removal with the understanding that the placement of a glaucoma drainage device might become necessary in the future.
On the day of surgery, I examined the patient once he was in a supine position. To my surprise, two implants were visible in the angle (Figure 5). I created a clear corneal incision and removed the implants (watch the video below). I then rotated my hand and performed an approximately 180º goniotomy enhancement with a straight Sinskey hook. I accessed the inferior angle through a superior incision. During the procedure, I visualized and removed a membrane that had covered the previous goniotomy site, resulting in good blood reflux (Figure 6).
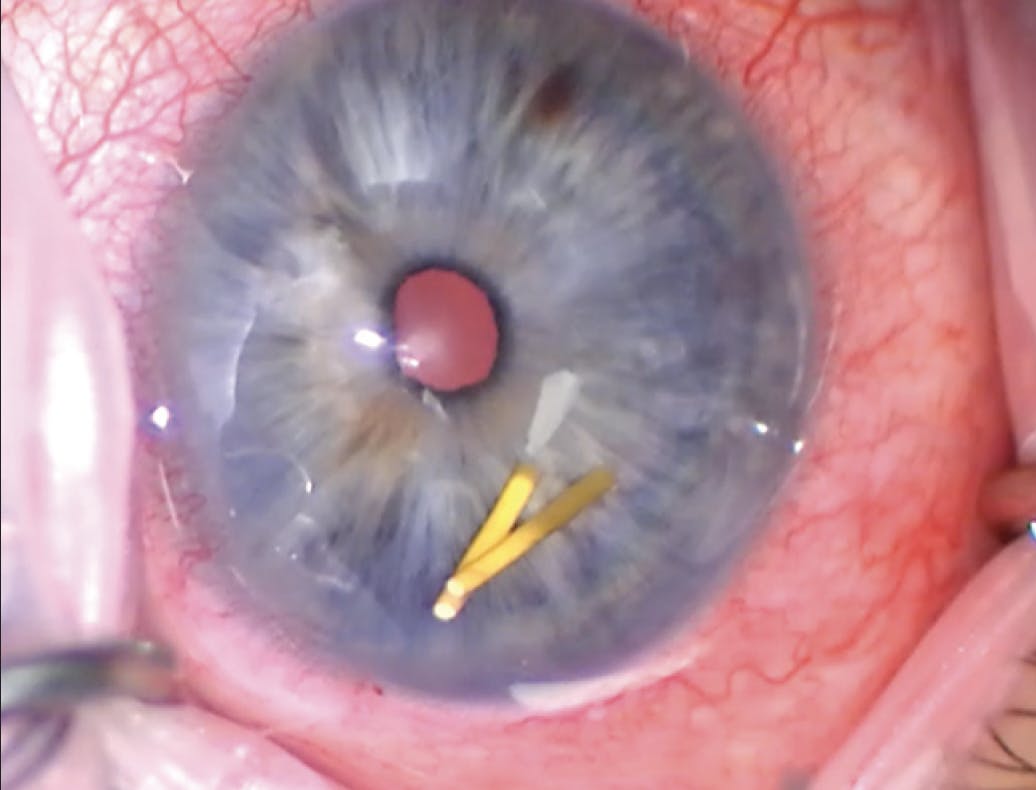
Figure 5. Surgical image of two fluocinolone acetonide intravitreal implants in the anterior chamber before removal.
On postoperative day 1, the patient’s visual acuity was hand motion, a diffuse microhyphema was evident, and the IOP was 45 mm Hg OD. Given that he was on maximal topical medical therapy, acetazolamide was added to his drug regimen. The following week, the IOP had decreased to 18 mm Hg, and his visual acuity was 20/30 OD. In the ensuing months, he was weaned off acetazolamide, timolol, and netarsudil.
At the patient’s last visit, 3 months after surgery, his UCVA was 20/20 OU. His IOP was 11 mm Hg OD and 9 mm Hg OS. His ocular hypotensive medications consisted of a fixed combination of brinzolamide and brimonidine tartrate ophthalmic solution in the left eye and latanoprost ophthalmic solution in both eyes. The right eye was quiet and required no steroid treatment.
1. Bollinger K, Kim J, Lowder CY, Kaiser PK, Smith SD. Intraocular pressure outcome of patients with fluocinolone acetonide intravitreal implant for noninfectious uveitis. Ophthalmology. 2011;118(10):1927-1931.
2. Burkholder BM, Wang J, Dunn JP, Nguyen QD, Thorne JE. Postoperative outcomes after fluocinolone acetonide implant surgery in patients with birdshot chorioretinitis and other types of posterior and panuveitis. Retina. 2013;33:1684-1693.
3. Goldstein DA, Godfrey DG, Hall A, et al. Intraocular pressure in patients with uveitis treated with fluocinolone acetonide implants. Arch Ophthalmol. 2007;125(11):1478-1485.
4. Mahmud H, Stewart JM. Migration of a fluocinolone acetonide implant (Yutiq) to the anterior chamber and its nonurgent removal. Am J Ophthalmol Case Rep. 2020;20:100987.
5. Xiao J, Zhao C, Liang A, Zhang M, Cheng G. Efficacy and safety of high-energy selective laser trabeculoplasty for steroid-induced glaucoma in patients with quiescent uveitis. Ocul Immunol Inflamm. 2021;29(4):766-770.
6. Cho J, Hogan D, Salim M, et al. Comparison of outcomes for laser trabeculoplasty after Kahook Dual Blade goniotomy versus in goniotomy-naive eyes. Ophthalmol Ther. 2021;10(4):905-912.
7. Morales J, Al Shahwan S, Al Odhayb S, Al Jadaan I, Edward DP. Current surgical options for the management of pediatric glaucoma. J Ophthalmol. 2013;2013:763735.












