Due to the progressive nature of glaucoma, when evaluating a treatment’s efficacy for patients, we clinicians must consider its long-term safety and duration of effect. To set clear expectations for ourselves and our patients, we rely on clinical trial data to support our prescription decisions.
One such clinical trial is showing promise for a unique formulation and delivery of travoprost, a prostaglandin analog used to reduce intraocular pressure (IOP) in patients with open-angle glaucoma or ocular hypertension. The iDose TR (travoprost intracameral implant) 75 mcg (Glaukos Corporation, Figure 1), a procedural pharmaceutical, is implanted directly into the eye where it is designed to steadily release a concentrated medication for up to 3 years, thus potentially offering an alternative to daily eye drop treatments.*
The iDose TR met its primary efficacy endpoint—noninferiority to topical timolol through 3 months—for FDA approval in late 2023. Phase 3 data also show iDose TR demonstrated 1.3 mm Hg statistically superior IOP-lowering versus pretrial prostaglandin analogs (PGAs).1
A Look at the Data
The 154-subject, multicenter, randomized, double-blind, phase 2b trial evaluated a single administration of iDose TR compared to twice-daily topical timolol ophthalmic solution 0.5%—the latter equating to approximately 2,190 eye drops per eye over the 3-year evaluation period.2
The data showed that 92% of patients in the iDose TR treatment group had well-controlled IOP with the same or fewer medications at 1 year, 72% at 2 years, and 69% remained relatively stable on the same or fewer medications at 3 years (Figure 2). By comparison, just 46% of subjects in the timolol control arm met the same benchmark. Further, iDose TR subjects performed similarly to timolol subjects at 36 months in terms of mean IOP reduction, yet with fewer topical medications versus timolol.2
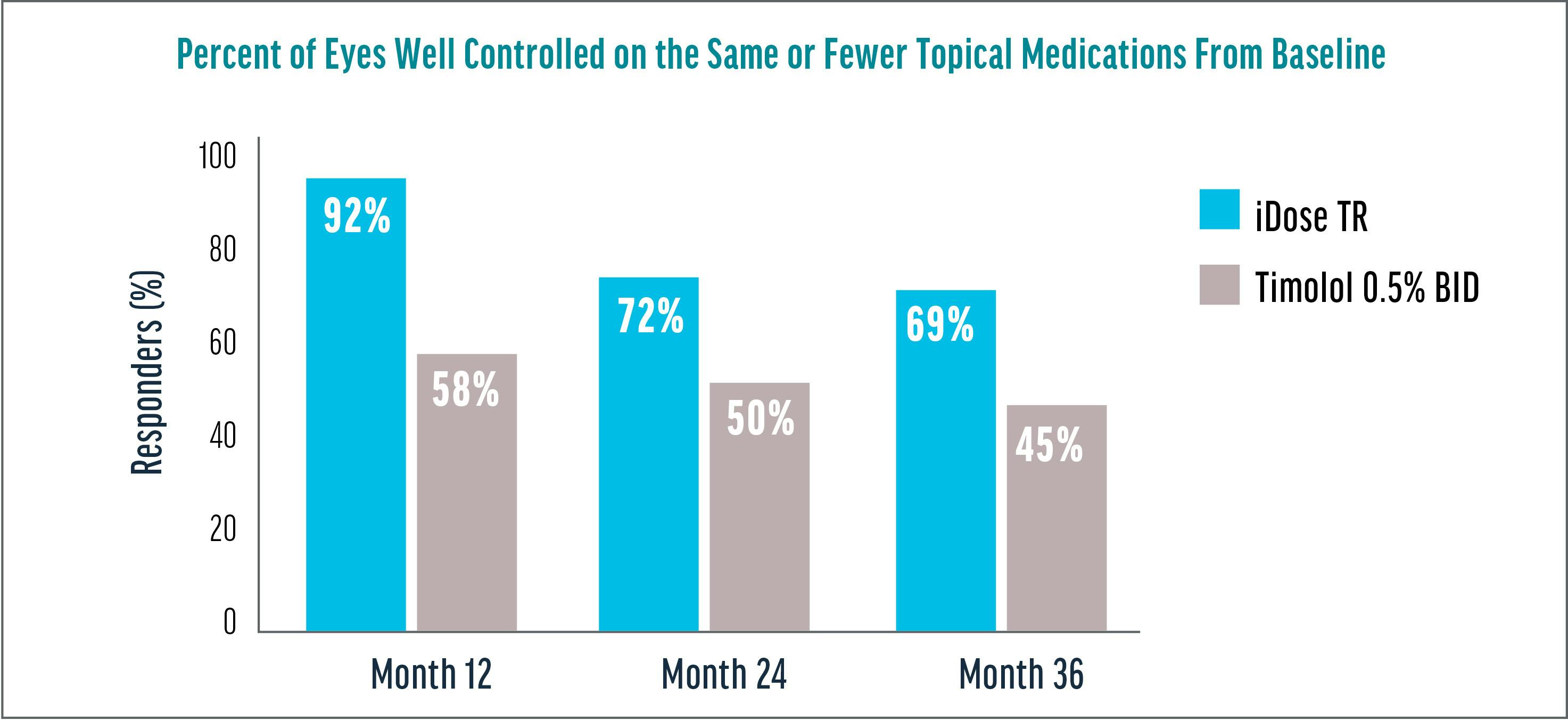
Figure 2. Three-year data show 69% of eyes are well controlled on the same or fewer topical medications from baseline.2†
• †Clinically and statistically relevant IOP-lowering effects were observed through month 36 with mean IOP reductions ranging from 7.3 to 8.0 mm Hg for iDose TR.
• Baseline was 24.82 mmHg.
• Well controlled was defined as pressures of 18 mm Hg and below.
Demonstrated Safety
iDose TR is made from an inert material that remains stable during the implantation procedure. With any implant, surgeons must ensure that the patient’s endothelial cells will not be compromised in any way. The phase 2b data demonstrated a favorable safety profile for iDose TR, with no clinically significant corneal endothelial cell loss, no serious corneal adverse events, and no adverse events of periorbital fat atrophy and conjunctival hyperemia reported to date in either elution arm.1
I find it reassuring to see that, in the phase 2b data, there was no clinically significant corneal endothelial cell loss, and I believe that is due to the material, the proprietary travoprost oil formulation, and the stability of the implant as it anchors into the scleral wall through the trabecular meshwork.
PHYSICIAN Confidence
It gives me greater confidence as a physician to proceed with a treatment with such sustained efficacy as shown through long-term data. The responder rate for the iDose TR is reassuringly high. The phase 2b data showed that IOP was well controlled at 1 year for 92% of patients, giving ophthalmologists the confidence to go into the operating room with the validation that the patient has a great chance of getting a good response.
Real-World Versus Study
In my practice, patients implanted with iDose TR have experienced positive results, and I find the implantation procedure to be highly intuitive, which eases my comfort level.
I recently treated a patient in whom I performed cataract surgery and iDose TR implantation. With the data from the phase 2b trial, I felt very comfortable with this approach from an inflammation and safety standpoint, knowing the outcomes of the trial showed low incidence of inflammation. These data open up our options as glaucoma surgeons, and give flexibility to use this drug delivery implant in a wide range of patients with OAG or OHT.
Removing the Drop Burden
I cannot overemphasize the impact of getting patients off topical IOP medications. From a clinical perspective, the overwhelming majority of my patients have compliance issues related to topical medications. Data from the phase 3 trial showed that, at 12 months, 81% of iDose TR patients were completely free of IOP-lowering topical medications.1 When I can tell a patient that I can decrease their burden even by just one fewer drop per day, it is a game changer, not just for their immediate quality of life but for more effectively managing their disease. iDose TR demonstrated 1.3 mm Hg statistically superior IOP-lowering versus pretrial PGAs,1 making it a “meaningful addition to the interventional glaucoma armamentarium." It addresses the disadvantages of topical IOP-lowering medications such as low adherence and topical side effects.2
I challenge colleagues to ask themselves: Are we doing the best for our patients by keeping them on a topical drop if they are having compliance challenges and not receiving the treatment as required? iDose TR is an additional tool for glaucoma surgeons to help alleviate the medication burden for patients. We have the opportunity to implant an IOP-controlling procedural pharmaceutical with safe, sustained efficacy, and we have the data to back it up.
1. iDose TR Phase 3 Clinical Trials, data on file, Glaukos Corporation.
2. Berdahl JP, Sarkisian SR Jr, Ang RE, et al. Efficacy and safety of the travoprost intraocular implant in reducing topical IOP-lowering medication burden in patients with open-angle glaucoma or ocular hypertension. Drugs. 2024;84(1):83-97.
3. Szekely G, Katz LJ, Voskanyan LA, et al. Travoprost intraocular implant (iDose® TR) delivers therapeutically relevant and durable aqueous humor drug concentrations at 24 months and demonstrates safety of repeat implant administration. Paper presented at ARVO Annual Meeting; May 5-9, 2024; Seattle, WA.
INDICATIONS AND USAGE
iDose TR (travoprost intracameral implant) is indicated for the reduction of intraocular pressure (IOP) in patients with open angle glaucoma (OAG) or ocular hypertension (OHT).
IMPORTANT SAFETY INFORMATION
Dosage and Administration
For ophthalmic intracameral administration. The intracameral administration should be carried out under standard aseptic conditions.
iDose TR is contraindicated in patients with active or suspected ocular or periocular infections, patients with corneal endothelial cell dystrophy (e.g., Fuch’s Dystrophy, corneal guttatae), patients with prior corneal transplantation, or endothelial cell transplants (e.g., Descemet’s Stripping Automated Endothelial Keratoplasty [DSAEK]), patients with hypersensitivity to travoprost or to any other components of the product.
Warnings and Precautions
iDose TR should be used with caution in patients with narrow angles or other angle abnormalities. Monitor patients routinely to confirm the location of the iDose TR at the site of administration. Increased pigmentation of the iris can occur. Iris pigmentation is likely to be permanent.
Adverse Reactions
In controlled studies, the most common ocular adverse reactions reported in 2% to 6% of patients were increases in intraocular pressure, iritis, dry eye, visual field defects, eye pain, ocular hyperaemia, and reduced visual acuity.
Please see full Prescribing Information.
You are encouraged to report all side effects to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088. You may also call Glaukos at 1-888-404-1644.
Dr. Singh has been compensated by Glaukos for his participation.
© 2024 Glaukos PM-US-2280


