Angle-closure glaucoma (ACG) accounts for 10% of all glaucoma in the United States. This form of the disease is more prevalent worldwide, however, with estimates that 5.3 million people will be blinded by primary ACG by 2020.1
ACG is caused by apposition of the peripheral iris to the trabecular meshwork, which reduces the drainage of aqueous humor through the anterior chamber angle. The principle cause of primary angle closure is pupillary block, whereas in secondary angle closure, there is physical blocking of the angle. Treatment depends on the type of angle closure present. Lowering IOP, if elevated, is always the first priority. This step is usually handled medically with topical therapy. If any degree of pupillary block is present or a potential threat, then a laser peripheral iridotomy (LPI) is indicated (Figure 1). Understanding when an LPI is indicated can help prevent delays in treatment as well as further complications.
INDICATIONS FOR LPI
Acute Angle Closure
Ninety percent of all US patients presenting with angle closure have pupillary block.2 Rapid reversal of the block provides relief to the patient in addition to minimizing the risk of vision loss and permanent damage to the angle leading to chronically elevated pressures. On examination, IOP is markedly elevated, and the angle is closed on gonioscopy (Figure 2). Initial therapy should be aimed at immediately lowering IOP with topical or oral medication and clearing the cornea. Osmotic agents such as glycerin used topically can aid in clearing the cornea quickly. Once the physician is able to visualize the iris, he or she should perform an LPI to relieve the pupillary block, deepen the angle, and lower the IOP. Relatively soon afterward, an LPI should be performed on the contralateral eye. Without a prophylactic LPI, the risk of an acute attack in the fellow eye may be as high as 51%.3
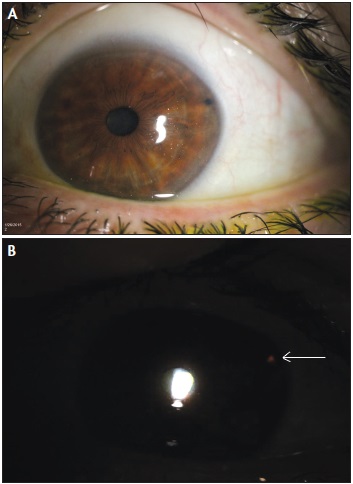
Figure 1. A temporally placed LPI (A). Transillumination confirms patency (B).
Occludable Angle
As mentioned earlier, any patient presenting with angle closure should be treated by means of an LPI, whereas a prophylactic LPI in individuals with a narrow angle reduces the chance of pupillary block and subsequent acute angle closure. Patients may present with symptoms of intermittent angle closure and complain of occasionally blurred vision, pain, or redness, often after the pupil dilated, such as in a dimly lit room. On gonioscopy, the angle appears occludable, meaning that the angle structures are not visible. There is no consensus on the threshold of when to perform an LPI based on a gonioscopic examination alone. Personally, if I can visualize less than two quadrants of trabecular meshwork, I proceed with an LPI. In symptomatic patients or those in whom indentation does not deepen the angle, an LPI is also likely indicated.
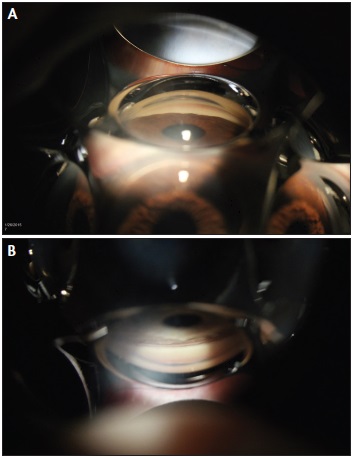
Figure 2. A narrow angle on gonioscopy with no angle structures visible (A). An open angle with an easily visualized scleral spur and ciliary body (B).
Narrow Anterior Chamber Angle
Patients with a narrow angle may be asymptomatic, but upon slit-lamp examination, the anterior chamber may appear shallow. Furthermore, on gonioscopy, the angle will appear narrow, and all angle structures likely will not be visible. In these cases, it is often difficult to determine if an LPI is necessary. An indication to proceed with an LPI may be if the patient has risk factors such as a family history of angle closure, an age over 60 years, female gender, and hyperopia.4 In patients at increased risk of angle closure, if an LPI is not implemented, a gonioscopic examination should be performed prior to any pupillary dilation to decrease the chance of inducing angle closure.
Chronic Angle Closure
Often, patients with long-standing, intermittent angle closure or narrow anterior chambers develop chronic appositional closure of the angle. In these cases, peripheral anterior synechiae may be visible on gonioscopy. The patient’s IOP will be elevated, but other signs of acute angle closure such as corneal edema or pain will be absent. In cases of chronic angle closure, an LPI relieves any pupillary block, but the chronicity of apposition means that the angle may not deepen due to synechiae formation. Nevertheless, an LPI is essential to prevent further pupillary block. Further laser or surgical intervention to reduce IOP may be needed.
Plateau Iris
Plateau iris is an anatomic variation in which the iris root angles forward from its insertion point and then again centrally.5 It is the only type of primary angle closure without pupillary block. In eyes with a plateau iris configuration, an LPI is indicated to relieve any component of pupillary block. The angle will often deepen after an LPI in these patients. In contrast, those with plateau iris syndrome will still have a narrow angle and increased pressure after an LPI. The procedure is still performed to make the definitive diagnosis of plateau iris syndrome versus configuration, but for the latter, further treatment such as laser iridoplasty may be needed to open the angle.
Aqueous Misdirection
Patients with aqueous misdirection develop a shallow anterior chamber due to the lens iris diaphragm’s being pushed anteriorly by the trapped aqueous in the posterior chamber. This condition leads to a pupillary block that can be relieved by an LPI. Additionally, further treatment for aqueous misdirection often involves disruption of the hyaloid face with an Nd:YAG laser, and the presence of a peripheral iridotomy in phakic patients is a prerequisite to be able to perform this procedure.
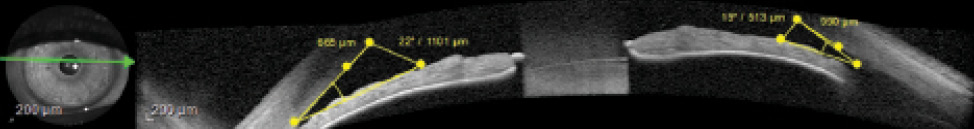
Figure 3. Anterior segment optical coherence tomography measuring the angle of insertion and demonstrating a narrow angle approach.
Pigment Dispersion Syndrome
The abnormal iris configuration in pigment dispersion syndrome leads to a reverse pupillary block. The iris bows posteriorly, chafing the iris against the lens zonules and releasing pigment that can block the trabecular meshwork. An LPI may relieve the posteriorly bowed iris configuration, decrease the release of pigment, and prevent further progression of pigment dispersion syndrome.
Secondary Angle Closures With Pupillary Block
Patients with secondary angle closure have an identifiable cause of iridocorneal apposition. Addressing the underlying cause with procedures such as cataract extraction offers definitive treatment but also carries the risks involved with surgery. In such cases, an LPI performed prior to other surgical intervention can decrease the risk of complications during surgery or relieve symptoms of acute angle closure and postpone the need for surgery. Many secondary angle closures are lens induced. Examples that may benefit from a therapeutic or prophylactic LPI are phacomorphic glaucoma, microspherophakia, and ectopia lentis.6-8
Uveitic eyes may also develop pupillary block secondary to fibrotic membranes or peripheral synechiae. The pupil may be scarred to the anterior capsule, blocking aqueous flow to the anterior chamber and leading to an iris bombé formation. An LPI is indicated to restore the proper flow of aqueous, deepen the chamber, and decrease pressure. Caution is warranted, however, because the procedure may exacerbate ocular inflammation.
ADDITIONAL TESTS TO DETERMINE LPI CANDIDATES
When the chamber appears to be shallow but the gonioscopic examination is inconclusive, other modalities or tests can aid diagnosis and treatment. Three different provocative tests have mostly fallen out of favor. A modification I have used is to dilate the pupil with tropicamide alone and recheck the IOP 1 hour later. I consider an increase of more than 8 mm Hg from baseline to be a positive test result and a risk factor for acute angle closure. I strongly consider an LPI for these patients.
A more objective test is anterior segment optical coherence tomography (AS-OCT). This technology can measure anterior chamber depth and the degree of the iridocorneal angle9,10 (Figure 3). Some studies have even shown that, after pupillary dilation, the iris is measurably thicker in patients with narrow angles, and AS-OCT may be able to detect this change.11 After LPI, deepening of the anterior chamber and angle is usually noted on slit-lamp examination, gonioscopy, and even AS-OCT.
CONTRAINDICATIONS
In secondary angle closure without pupillary block, outflow is obstructed due to direct blockage of the trabecular meshwork, as in neovascular glaucoma and iridocorneal endothelial syndrome. An LPI is not indicated in these cases. In drug-induced angle closure, treatment is cessation of the causative agent; an LPI is not indicated.
CONCLUSION
An LPI is often indicated in patients with angle closure. This procedure can treat pupillary block or prevent it from occurring. Gonioscopy and knowledge of angle anatomy are fundamental to identifying patients with angle closure, and an understanding of the etiology of narrowing is essential to determining which patients may benefit from an LPI. n
Shuchi Patel, MD, is director of Glaucoma Services and an assistant professor at Loyola University Medical Center in Maywood, Illinois. Dr. Patel may be reached at shuchibpatel@gmail.com.
1. Quigley HA, Broman AT. The number of people with glaucoma worldwide in 2010 and 2020. Br J Ophthalmol. 2006;90(3):262-267.
2. Ritch R, Chang BM, Liebmann JM. Angle closure in younger patients. Ophthalmology. 2003;110:1880-1889.
3. Henrietta H, Chew P, Sng C, et al. A comparison of two approaches to managing acute primary angle closure in Asian eyes. Clin Ophthalmol. 2013;7(7):1205-1210.
4. See JLS, Chew PTK. Angle-closure glaucoma. In: Yanoff M, Duker JS, eds. Ophthalmology. 3rd ed. Philadelphia, PA; Mosby; 2009:1162.
5. Diniz F, Cronemberger S, Merula RV, Calixto N. Plateau iris. Arg Bras Oftalmol. 2008;71(5):752-758.
6. Kaplowitz K, Kapoor K. An evidence-based approach to phacomorphic glaucoma. J Clinic Exp Ophthalmol. 2012;S1:006. http://omicsonline.org/2155-9570/2155-9570-S1-006.digital/2155-9570-S1-006.html. Accessed March 16, 2015.
7. Senthil S, Rao HL, Hoang NT, et al. Glaucoma in microspherophakia: presenting features and treatment outcomes. J Glaucoma. 2014;23(4):262-267.
8. Tsilou E, MacDonald IM. Weill-Marchesani syndrome. In: Pagon RA, Adam MP, Bird TD, et al, eds. GeneReviews. Seattle, WA: University of Washington, Seattle; 1993-2013.
9. Wong HT, Lim MC, Sakata LM, et al. High-definition optical coherence tomography imaging of the iridocorneal angle of the eye. Arch Ophthalmol. 2009;127:256.
10. Guzman CP, Gong T, Nongpiur ME, et al. Anterior segment optical coherence tomography parameters in subtypes of primary angle closure. Invest Ophthalmol Vis Sci. 2013;54:5281.
11. Quigley HA, Silver DM, Friedman DS, et al. Iris cross-sectional area decreases with pupil dilation and its dynamic behavior is a risk factor in angle closure. J Glaucoma. 2009;18:173.
