Primary open-angle glaucoma is the most common form of the disease in the United States, and a subset of patients have normal-tension glaucoma. Current glaucoma treatment aims to lower IOP through medical therapy or surgery. Although decreasing IOP slows disease progression, retinal ganglion cell (RGC) loss and glaucomatous degeneration may continue.1
Novel therapies are clearly needed to replace lost outflow tract cells and to protect and perhaps even replace RGCs. Human stem cells have shown promise and deserve attention, not just in the laboratory, but in the clinical setting as well. This article provides an overview of stem cells for the treatment of glaucoma via neuroprotection, neuroenhancement, and possibly cell replacement strategies.
ONGOING CLINICAL TRIALS
US clinical trials have begun to investigate how neuroprotective therapies may help slow or even prevent RGC degeneration. For example, researchers assessed encapsulated cell implants secreting ciliary neurotrophic factor for safety, preserving vision (neuroprotection), and improving vision (neuroenhancement) in patients with severe glaucoma (Figure 1).2 Although stem cells were not used in this trial and the results have not yet been published, this research is indicative of the field's movement towards new cell-based neuroprotective therapeutics that avoid multiple intravitreal injections.3,4 Preclinical data have suggested that mesenchymal stem cells such as those derived from bone marrow are neuroprotective when transplanted in rats.5,6
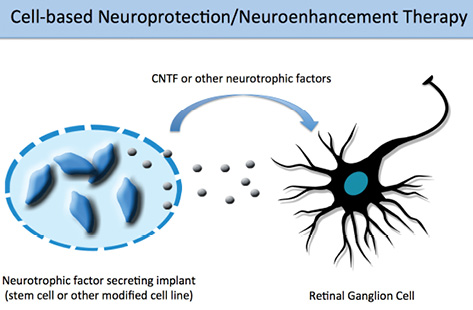
Figure 1. Neurotrophic factors may be secreted by stem cells or other modified cell lines that can either be injected directly into the eye or placed in a semipermeable capsule. These neurotrophic factors may have neuroprotective and/or neuroenhancing effects on RGCs, thus preserving vision and perhaps improving cellular function. Abbreviation: CNTF, ciliary neurotrophic factor.
APPROACHES TO STEM CELL-DERIVED THERAPIES IN GLAUCOMA
Most clinical trials using stem cells have focused on neuroretinal degenerative diseases other than glaucoma, and it is not surprising why. Once the optic nerve begins to degenerate in glaucoma, many barriers must be overcome in order to functionally replace these RGCs. Once implanted, a stem cell-derived RGC would need to migrate to the ganglion cell layer, partner with the appropriate neuroretinal cells, extend axons to the optic nerve head and along the nerve itself, and finally synapse with the correct visual targets in the brain. Although the goal of successfully replacing dead RGCs is unlikely to be reached in the immediate future, preclinical studies of stem cells for cell replacement therapy in the treatment of glaucoma, including optic nerve regeneration, are underway.7
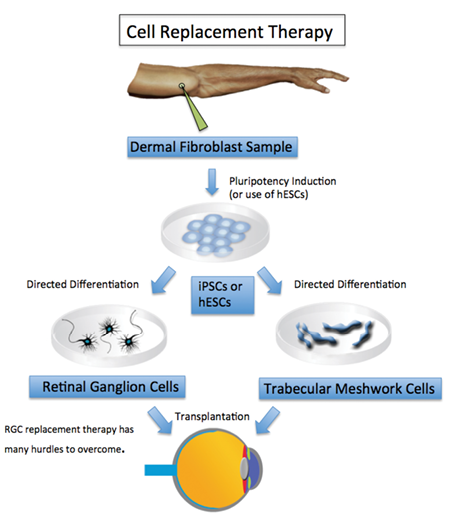
Figure 2. In the treatment of glaucoma, cell replacement therapy would involve the directed differentiation of stem cells (human embryonic [hESCs] or iPSCs) to the appropriate ocular cell type for transplantation. In this image, stem cells are directed to either TM cells or RGCs for novel therapies that would help maintain aqueous outflow or replace the damaged RGCs, respectively.
Trabecular meshwork (TM) cell replacement may hold more immediate promise for translation to clinical use. Recently, investigators used human anterior segments in an ex vivo perfused outflow pathway organ culture model to see if stem cells could restore homeostatic function after saponin-induced TM cell loss.8 Human induced pluripotent stem cells (iPSCs) were differentiated into TM-like cells and transplanted into the organ culture model, thereby restoring aqueous outflow. It is exciting to imagine that TM cell replacement by patient-derived TM-like cells may one day help physicians to treat patients whose IOP is elevated due to a loss of functional cells in the aqueous outflow tract.
STEM CELL THERAPIES IN OTHER RETINAL DEGENERATIVE DISEASES
Stem cell-derived tissue replacement therapy for other retinal degenerative diseases is already in human clinical trials. In September 2014, a Japanese trial at RIKEN made history with the first human iPSC-derived tissue transplantation ever, which took place in the eye. An autologous iPSC-derived sheet of retinal pigment epithelial cells was surgically implanted in a patient with age-related macular degeneration.9
An update on two cell replacement trials for patients with Stargardt disease and age-related macular degeneration was published recently.10-12 After subretinal implantation of human embryonic stem cell-derived retinal pigment epithelial cells, patients were observed for an average of 22 months. BCVA improved in 10 eyes and improved or remained unchanged in seven eyes. Only one eye experienced vision loss. Importantly, there were no adverse outcomes due to the transplant.
FUTURE DIRECTIONS
Over the next few years, stem cell-derived glaucoma therapies will likely progress in the areas of RGC neuroprotection and neuroenhancement as well as TM cell replacement, all aimed at halting vision loss and perhaps recovering vision (Figure 2). As for pushing the boundaries of stem cell science with RGC replacement and sight restoration, it seems unlikely that the field will advance to human subjects in the near future. That said, only 8 years passed between Yamanaka's ground-breaking generation of iPSCs and the first transplantation of autologous iPSC-derived retinal tissue into the human eye.9,13 All in all, however, given the complexities of RGC replacement and optic nerve regeneration, the restoration of vision lost to glaucoma will likely require an interdisciplinary approach involving advances in stem cell biology, neuroscience, and tissue engineering. n
The authors thank Jonathan Simon Green for his artistic contribution in creating Figures 1 and 2.
David I. Green, BS, is a fourth-year medical student at the University of California, San Francisco. Mr. Green may be reached at david.green@ucsf.edu.
Yvonne Ou, MD, is a glaucoma specialist and assistant professor at the University of California, San Francisco. Dr. Ou may be reached at yvonne.ou@ucsf.edu.
1. The AGIS Investigators. The Advanced Glaucoma Intervention Study (AGIS): 7. The relationship between control of intraocular pressure and visual field deterioration. Am J Ophthalmol. 2000;130(4):429-440.
2. Goldberg JL. NT-501 CNTF implant for glaucoma: safety, neuroprotection and neuroenhancement: NCT01408472. Clinicaltrial.gov. https://clinicaltrials.gov/ct2/show/NCT01408472?term=NCT01408472&rank=1. Updated December 25, 2014. Accessed March 25, 2015.
3. Johnson TV, Bull ND, Hunt DP, et al. Neuroprotective effects of intravitreal mesenchymal stem cell transplantation in experimental glaucoma. Invest Ophthalmol Vis Sci. 2010;51:2051-2059.
4. Ng TK, Fortino VR, Palaez D, et al. Progress of mesenchymal stem cell therapy for neural and retinal diseases. World J Stem Cells. 2014;6(2):111-119.
5. Hu Y, Tan HB, Wang XM, et al. Bone marrow mesenchymal stem cells protect against retinal ganglion cell loss in aged rats with glaucoma. Clin Interv Aging. 2013;8:1467-1470.
6. Yu S, Tanabe T, Dezawa M, et al. Effects of bone marrow stromal cell injection in an experimental glaucoma model. Biochem Biophys Res Commun. 2006;344:1071-1079.
7. de Lima S, Koriyama Y, Kurimoto T, et al. Full-length axon regeneration in the adult mouse optic nerve and partial recovery of simple visual behaviors. Proc Natl Acad Sci USA. 2012;109:9149-9154.
8. Abu-Hassan DW, Li X, Ryan EI, et al. Induced pluripotent stem cells restore function in a human cell loss model of open-angle glaucoma. Stem Cells. 2015;33(3):751-761.
9. Foundation for Biomedical Research and Innovation RIKEN. First RPE cell sheet graft transplant. http://www.riken.jp/en/pr/topics/2014/20140912_1/. September 12, 2014. Accessed March 24, 2015.
10. Ocata Therapeutics. Sub-retinal transplantation of hESC derived RPE (MA09-hRPE) cells in patients with Stargardt's macular dystrophy: NCT01345006. Clinicaltrial.gov. https://clinicaltrials.gov/ct2/show/NCT01345006?term=NCT01345006&rank=1. Updated November 3, 2014. Accessed March 25, 2015.
11. Ocata Therapeutics. Safety and tolerability of sub-retinal transplantation of hESC derived RPE (MA09-hRPE) cells in patients with advanced dry age related macular degeneration (dry AMD): NCT01344993. Clinicaltrial.gov. https://clinicaltrials.gov/ct2/show/NCT01344993?term=NCT01344993&rank=1. Updated November 3, 2014. Accessed March 25, 2015.
12. Schwartz SD, Regillo CD, Lam Bl et al. Human embryonic stem cell-derived retinal pigment epithelium in patients with age-related macular degeneration and Stargardt's macular dystrophy: follow-up of two open-label phase 1/2 studies. Lancet. 2015;385:509-516.
13. Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663-676.
