CASE PRESENTATION
An 82-year-old woman was being seen by her retinal specialist for follow-up for nonexudative macular degeneration in both eyes. Her past medical history was noncontributory, and her ocular history included cataract surgery on both eyes in 2008, chronic uveitis and chronic cystoid macular edema in the left eye since 2008, and glaucoma diagnosed in the left eye in 2010. Her ocular medications included dorzolamide 2% three times daily, travoprost 0.04% at night, fixed-combination timolol 0.5%-brimonidine 2% twice daily, and prednisolone 1% twice daily, all in the left eye. She complained of new symptoms in her left eye—increased floaters, decreased vision, and photophobia. The retinal physician referred the patient to our practice to determine if the prostaglandin could be stopped and for the consideration of other options for managing her IOP. Interestingly, to help determine the etiology of the patient’s recalcitrant inflammation that had persisted for the past 2 years, a complete uveitis workup had been performed by the referring physician, including laboratory testing and imaging that had essentially been unremarkable.
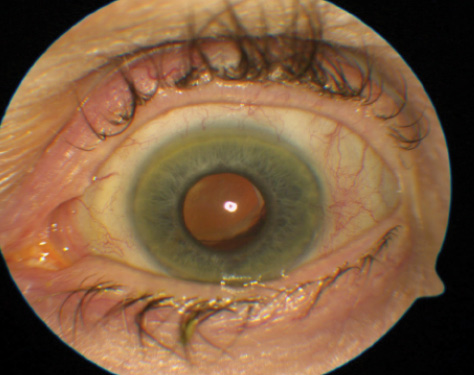
Figure 1. A slit-lamp photograph of the left eye shows mild superior decentration of the IOL with the inferior edge of the optic visible through a middilated pupil.
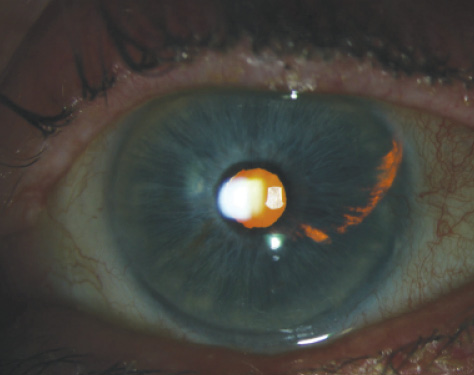
Figure 2. A slit-lamp photograph shows transillumination defects of the iris corresponding to the temporal haptic of the IOL.

Figure 3. A goniophotograph shows an inferior angle with excessive pigment and the presence of a microhyphema.
Upon presentation to the Glaucoma Service at the Doheny Eye Institute, the patient complained of redness, pain, and photophobia in her left eye. Her visual acuity was 20/25 OD and 20/60 OS. Her IOP was 17 mm Hg OD and 28 mm Hg OS. A slit-lamp examination of the right eye was unremarkable except for a three-piece silicone IOL in the sulcus. In the left eye, there were occasional pigmented cells in the anterior chamber and pigment deposition on the corneal endothelium. There was a one-piece acrylic IOL, with the optic mostly situated in the capsular bag; the nasal haptic was visible and within the capsular bag (Figure 1). The temporal haptic was outside the capsule and sitting in the ciliary sulcus with a notable overlying iris transillumination defect temporally (Figure 2). Gonioscopy revealed a grade 4 open angle with a small hyphema in the inferior angle (Figure 3). There was also 2+ to 3+ posterior capsular opacification and 1+ vitreous debris in the left eye. A fundus examination revealed macular drusen, changes in the retinal pigment epithelium, and some cystic elevation of the left fovea. The vertical cup-to-disc ratio was 0.4 with good rims in the right eye, and 0.75 with inferior rim thinning and a corresponding mild superior nasal step visual field defect on automated perimetry in the left eye. Optical coherence tomography confirmed cystoid macular edema in the left eye (Figure 4).
Based on the results of the examination, we suspected uveitis-glaucoma-hyphema (UGH) syndrome as a cause of secondary pigment dispersion and the patient’s persistent cystoid macular edema. The underlying mechanism was likely due to the chafing of the malpositioned IOL’s temporal haptic against the posterior pigmented iris epithelium.
HOW WOULD YOU PROCEED?
- Would you stop the prostaglandin and add pilocarpine?
- Would you examine the patient further?
- Would you obtain additional imaging to acquire more information?
- Would you reposition the IOL or remove and exchange it?

Figure 4. Visual field testing reveals mild to moderate glaucoma defects in the left eye. There is a generalized depression and a superior nasal step/early arcuate defect.
surgICAL COURSE
After discussing the diagnosis with the patient, we decided to attempt repositioning the IOL’s temporal haptic back into the capsular bag. We made a 2-mm limbal temporal incision and created a paracentesis superiorly. Using an endoscope, we visualized the temporal lens haptic displaced outside the capsule in the ciliary sulcus; it was in direct contact with the posterior iris. Our attempts to open the capsule temporally to allow repositioning of the temporal haptic from the sulcus cavity to the capsular bag were unsuccessful, because the anterior and posterior capsules had fully fibrosed together temporally. Using microscissors and microforceps (MicroSurgical Technology), we decided to cut and remove the temporal haptic instead. The optic and nasal haptic were left untouched and sitting within the capsular bag. Given the patient’s degree of glaucoma and recent history of elevated and uncontrolled IOP despite maximally tolerated medical therapy, we also placed a Baerveldt 350 glaucoma implant (Abbott Medical Optics) in the superior temporal quadrant.
OUTCOME
The patient did well postoperatively. Her IOP in the operated eye improved (range, 10-13 mm Hg), and she was able to discontinue the dorzolamide and travoprost. One month postoperatively, the uveitis and hyphema had totally resolved, and her visual acuity had improved to 20/30. At the 2-year postoperative visit, the patient required three glaucoma medications to maintain her target IOP. Fortunately, she has maintained 20/25 visual acuity and has had no recurrences of uveitis.
DISCUSSION
UGH syndrome was first described in 1978 as a complication of Choyce-style anterior chamber IOLs due to the lenses chafing the iris.1 It is believed that excessive movement of the lens on the iris’ surface breaks down the blood-aqueous barrier. UGH syndrome is also a potential complication of posterior chamber sulcus lenses.2 There have even been reported cases of UGH syndrome secondary to a posterior chamber IOL that is fully enclosed in the capsule.3 Foroozan et al described a case in which there was intracapsular fixation of both haptics, but in which there was also proximity of the edge of the optic to the inferior pupillary margin in the region of an iridociliary body cyst. During miosis, there was nearly direct apposition of the lens optic with the iris cyst. The authors hypothesized that the cyst might have also changed the orientation of the zonules, causing more anterior movement of the lens-capsular apparatus.
Classic clinical features of UGH syndrome include hyphema, pigment-like dispersion in the anterior chamber, pigment on the corneal endothelium, iris transillumination defects, and a chronic fluctuation of symptoms. In some cases, patients experience transient visual obscurations similar to those of amaurosis fugax. The differential diagnoses include uveitic glaucoma, pigment dispersion syndrome, Possner-Schlossman syndrome, and hyphema secondary to another cause (neovascular glaucoma, iris neoplasia, etc.). An important element of making the correct diagnosis involves examining the undilated pupil for transillumination defects and evaluating the IOL’s position. Gonioscopy can help clinicians identify a microhyphema or any other causes of bleeding such as neovascular glaucoma. Another useful diagnostic tool is ultrasound biomicroscopy, which can help the clinician visualize the anterior segment (particularly posterior to the iris) and can reveal the underlying pathophysiology.4 Using radial and transverse sections on ultrasound biomicroscopy, the positioning of the haptics and optics and their relationships to the surrounding structures can be determined.
When possible with modern cataract surgery, inserting the IOL into the native capsular bag is generally preferred.5 It is also recommended that one-piece IOLs (with their intrinsically thicker haptics) not be placed in the sulcus so as to avoid risks of secondary pigment dispersion. In situations where there is a need for a sulcus-positioned IOL, such as in cases of a compromised posterior capsule, it is generally recommended that three-piece IOLs (with their intrinsically thinner haptics) be used.5 If this strategy is not an option, other approaches include a sclera-fixated IOL posterior to the iris, an iris-fixated IOL posterior to the iris, or an anterior chamber IOL. We considered these options for our case, but on endoscopic visualization, the lens optic appeared to be in a stable position within a fibrotic capsule. The haptic was cut safely with minimal disruption to the remainder of the lens. Another option was to remove and exchange the lens. Previous studies have reported an overall 72% success rate for IOL explantation and exchange for any cases needing IOL exchange.6
When UGH syndrome causes elevated IOP that is refractory to medications, many surgical approaches can be taken. If the presentation is very recent, one can simply reposition the IOL and monitor the patient for any improvement in IOP and inflammation while concurrently managing him or her medically as needed. Another approach that is especially suited to patients with advanced glaucoma who cannot tolerate a high IOP for a long period of time is to combine a lens procedure (repositioning or exchange) with glaucoma surgery. Nonfiltering options such as a trabecular bypass microstent (iStent; Glaukos) or ab interno trabeculotomy (Trabectome; NeoMedix) could be considered. In a patient with very high IOP or advanced glaucoma, however, a more aggressive treatment such as filtering surgery may be indicated. In an acutely inflamed eye, it is thought that trabeculectomy may be a more challenging surgery and more likely to fail early due to excessive tissue scarring. A glaucoma drainage device may be more likely to be effective and maintain the decrease in IOP long term. As with every case, care needs to be individualized and customized to a patient’s particular needs. n
Sourabh Arora, MD, is a resident in the Department of Ophthalmology at the University of Toronto, Canada. He acknowledged no financial interest in the products or companies mentioned herein. Dr. Arora may be reached at sourabh.arora@medportal.ca.

Vikas Chopra, MD, is a glaucoma specialist at Doheny Eye Center and an associate professor of ophthalmology at the David Geffen School of Medicine, University of California, Los Angeles. He acknowledged no financial interest in the products or companies mentioned herein. Dr. Chopra may be reached at vchopra@doheny.org.

Brian A. Francis, MD, MS, is a glaucoma specialist Doheny Eye Center and a professor of ophthalmology at the David Geffen School of Medicine, University of California, Los Angeles. He acknowledged no financial interest in the products or companies mentioned herein. Dr. Francis may be reached at bfrancis@doheny.org.
1. Choyce DP. Complications of the AC implants of the early 1950s and the UGH or Ellingson syndrome of the late 1970s. J Am Intraocul Implant Soc. 1978;4(2):22-29.
2. Aonuma H, Matsushita H, Nakajima K, et al. Uveitis-glaucoma-hyphema syndrome after posterior chamber intraocular lens implantation. Jpn J Ophthalmol. 1997;41:98-100.
3. Foroozan R, Tabas JG, Moster ML. Recurrent microhyphema despite intracapsular fixation of a posterior chamber intraocular lens. J Cataract Refract Surg. 2003;29:1632-1635.
4. Piette S, Canlas OA, Tran HV, et al. Ultrasound biomicroscopy in uveitis-glaucoma-hyphema syndrome. Am J Ophthalmol. 2002;133:839-884.
5. Chang DF, Masket S, Miller KM, et al; ASCRS Cataract Clinical Committee. Complications of sulcus placement of single-piece acrylic intraocular lenses. J Cataract Refract Surg. 2009;35(8):1445-1458.
6. Sinskey RM, Amin P, Stoppel JO. Indications for and results of a large series of intraocular lens exchanges. J Cataract Refract Surg. 1993;19:68-71.
