


New diagnostic modalities for glaucoma include confocal scanning laser ophthalmoscopy (CSLO; HRT [Heidelberg Engineering]), scanning laser polarimetry (SLP; GDx VCC [Carl Zeiss Meditec]), and optical coherence tomography (OCT). These new quantitative technologies provide seemingly “hard, objective” printouts suggesting both diagnosis and change. Given the importance of the structural evaluation of the optic nerve and retina in glaucoma, the rapid acceptance and widespread use of these expensive techniques is understandable, although perhaps not justifiable. We question whether they are as appropriate as color photographs of the optic nerve for evaluating patients with glaucoma or suspected of having the disease.
AT A GLANCE
• The two purposes of obtaining an image of the optic nerve head and/or retina when evaluating patients with or suspected of having glaucoma are (1) to diagnose them with the condition and (2) to assess disease progression.
• Advanced imaging technology does not permit the assessment of color, pallor, disc hemorrhages, narrowing of the retinal arteries, dilation of the retinal veins at the disc margin, peripapillary atrophy, texture of the rim tissue, and the course of the blood vessels, all of which are useful in diagnosis.
• A photograph taken 100 years ago remains useful, because the newer image uses essentially the same methodology. In contrast, the constant evolution of advanced imaging technology means that new images can no longer be validly compared to the image obtained at baseline, because the latest methods may not be compatible with older ones.
PURPOSES OF IMAGING THE OCULAR FUNDUS
The two purposes of obtaining an image of the optic nerve head and/or retina when evaluating patients with or suspected of having glaucoma are (1) to diagnose them with the condition and (2) to assess disease progression.
The diagnosis of glaucoma is complicated by the fact that almost every finding of the disease also occurs in people without the condition. What needs to be sought, then, are signs unique to glaucoma. These pathognomonic findings include
- focal pallor and narrowing of the rim so that the rim in that area is less than 0.1 rim/disc ratios wide
- an acquired pit of the optic nerve
- rim/disc asymmetry not from a cause such as asymmetry of disc size
- a characteristic pattern of nerve fiber bundle loss associated with a related field loss
- an IOP greater than approximately 35 mm Hg
- a rate of change in disc and field greater than what occurs with normal aging
There is a qualitative aspect to all of these findings. In this regard, photographs are superior to OCT, CSLO, or SLP, none of which allows the assessment of color and pallor and none of which is as helpful in evaluating other clues such as disc hemorrhages, narrowing of the retinal arteries, dilation of the retinal veins at the disc margin, peripapillary atrophy, texture of the rim tissue, and course of the blood vessels. No thresholds have been established for measurements such as retinal nerve fiber layer (RNFL) thickness: a thin RNFL (eg, 60 µm) is not a valid sign of glaucoma, whereas a notch associated with local pallor is a sure indication of the disease.
ASSESSMENT OF CHANGE
A valid determination of change demands an accurate baseline against which future measurements can be compared. Based on a study of OCT, CSLO, and disc photographs taken in our practice’s office, a major proportion of OCT measurements (often more than one-third) have a signal strength that is not high enough to ensure the validity of the image.1 One cause of poor signal strength is ocular surface disease (present in about 60% of long-term users of glaucoma medications).2 The result is artifactually thinned estimates of RNFL thickness. Such images are useless as baselines. This is not a problem with photographs.
Also disturbing, as new technology is introduced, which is constantly happening with OCT, CSLO, and SLP devices, the new image can no longer be validly compared to the baseline. This is not a problem with photographs: one taken 100 years ago is still useful.
COST AND CONVENIENCE
The costs passed on to the patient for images obtained with a fundus camera, a spectral-domain OCT device, the GDx VCC, and the HRT 3 are roughly the same. The fundus camera requires virtually no upkeep and will last 50 or more years, whereas the other three devices require expensive upkeep and will probably be obsolete in 5 years. In today’s world, most homeowners only live in the same locality for around 13 years.3 When they move, their previous fundus image is useless unless acquired using the same modality; this requirement is easy to meet with disc photography and extremely difficult to fulfill with all of the other imaging techniques.
CHARACTERISTICS
Qualitative Versus Quantitative
A great advantage of disc photographs is that they are the only imaging methodology that provides both qualitative and quantitative information. The pattern of change often reveals more than the amount of change.
Objective Versus Subjective
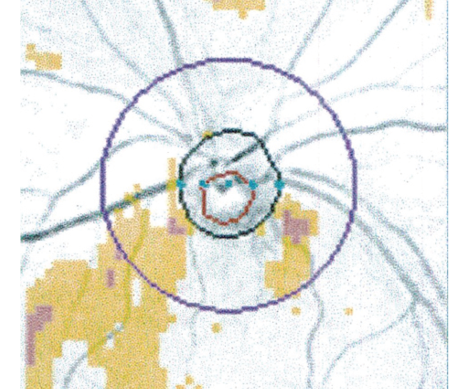
Figure. OCT deviation map. The red line shows the distance from optic disc margin to the peripapillary nerve fiber layer calculation circle with a fixed diameter of 3.4 mm centered on the optic disc.
OCT, CSLO, and SLP provide printouts that the machine interprets. This “objective” analysis often fails to take into account variabilities such as the difference in RNFL thickness associated with myopia or distance from the edge of the optic nerve head. The region scanned is a standard distance from the center of the optic disc; because the distance between the outer edge of the disc and the area scanned (red line in Figure) is much greater with small discs than large ones, the latter will incorrectly seem to have a thicker RNFL than the former, but the printout does not indicate that. All the examiner sees is an apparently hard figure (eg, 100 µm). Yes, it is hard, but it is not necessarily valid.4-6
It is therefore much easier to think that what OCT, CSLO, and SLP devices show is more valid than what disc photographs reveal, because observers interpret the latter. Eye care providers tend to forget the subjective component that came from a person’s instructing the machine to perform analysis in the manner that the programmer subjectively believes is best, but this is always done looking for the same data with a standard algorithm that may not apply to a particular disc being studied. This subjective requirement is actually one of the great advantages of photographs: they require thought. That thinking allows utilization of the best method of interpretation, hones skills, and promotes quality care.
THE APPARENT STATE OF THE ART
Offices offering OCT, CSLO, and SLP may seem to be more up to date, more “state of the art,” because they have the newest equipment, but being new does not make something the most artful. The appropriate art of medicine (ie, the proper goal of care) is to be as effective, convenient, useful, and affordable as possible. When those are the desired goals, disc photographs are still state of the art. n
1. Wu Z, Huang J, Dustin L, Sadda SR. Signal strength is an important determinant of accuracy of never fiber layer thickness measurements by optical coherence tomography. J Glaucoma. 2009;18(3):213-216.
2. Leung EW, Medeiros FA, Weinreb RN. Prevalence of ocular surface disease in glaucoma patients. J Glaucoma. 2008;17(5):350-355.
3. Emrath P. Latest study shows average buyer expected to stay in a home 13 years. NAHB website. Published January 3, 2013. http://bit.ly/1UZkmXY. Accessed May 13, 2016.
4. Garudadri CS, Rao HL, Parikh, RS, et al. Effect of optic disc size and disease severity on the diagnostic capability of glaucoma imaging technologies in an Indian population. J Glaucoma. 2012;21(7):475-480.
5. Marsh BC, Cantor LB, WuDunn D, et al. Optic nerve head (ONH) topographic analysis by Stratus OCT in normal subjects: correlation to disc size, age, and ethnicity. J Glaucoma. 2010;19(5):310-318.
6. Medeiros FA, Zangwill LM, Bowd C, et al. Influence of disease severity and optic disc size on the diagnostic performance of imaging instruments in glaucoma. Invest Ophthalmol Vis Sci. 2006;47(3):1008-1015.
George L. Spaeth, MD
• Louis Esposito research professor, Glaucoma Research Center, Wills Eye Hospital/Kimmel Medical College/Jefferson University, Philadelphia
• (215) 928-3123; gspaeth@willseye.org
• financial interest: none acknowledged
Kamran Rahmatnejad, MD
• glaucoma research fellow, Wills Eye Hospital, Philadelphia
• (215) 923-3128; krahmatnejad@willseye.org
• financial interest: none acknowledged
Lichuan Zeng, BSE
• glaucoma research assistant, Wills Eye Hospital, Philadelphia
• (215) 923-3128; lzeng78@hotmail.com
• financial interest: none acknowledged
