
The term artificial intelligence (AI) can be used to encompass big data analytics, deep learning, and robotics. Taken together, these technologies may include the process by which a computer gathers information (big data), analyzes it, and makes a decision in a process mimicking human cognition (J. Gordon and D. Tsay, unpublished data, 2017). A simple way to understand big data is that it usually includes data sets too large for commonly used software tools to capture, curate, manage, and process within a tolerable amount of time.
AT A GLANCE
• Big data is a powerful tool that can assist with patients’ care, improve outcomes, and deliver evidence-based and cost-effective health care.
• Big data is the basis of machine deep learning and artificial intelligence.
• Artificial intelligence is positioned to revolutionize medicine and provide new advances in health care.
In 2010, Eric Schmidt, then CEO of Google, said at the Techonomy conference in Lake Tahoe, California, “As much data is now being created every 2 days, as was created from the beginning of human civilization to the year 2003.”1 Similarly, the growth in structured and unstructured (difficult-to-use) medical data (imaging, electronic health records, etc.) has been exponential and has the potential to improve and transform health care delivery and outcomes. Data collection will not produce better results, however, unless the information is used intelligently. The marriage between big data and machine learning has the potential to improve health care outcomes through pattern recognition and deep learning algorithms.
IMPROVING HEALTH CARE
In her 2015 Edward Jackson Memorial Lecture, Anne Coleman, MD, PhD, stated that, with large enough data sets, it is possible to investigate rare events in cataract surgery so as to improve the procedure’s success rates by further decreasing the rate of complications. Dr. Coleman said, “Big data is a tool, and it’s really up to us how to use it.”2
The National Academy of Medicine has suggested in its learning health care systems workshop the integration of patients’ care with medical research through the use of electronic health records and registries. Health care practices could thus be continuously studied and improved.3 One example is the Intelligent Research in Sight (IRIS) Registry.
Launched by the American Academy of Ophthalmology in 2014, the IRIS Registry was designed as a longitudinal clinical data registry to store patients’ data. It performs statistical analysis to improve patients’ care and outcomes, develop benchmarks, deliver evidence-based and cost-effective health care, conduct and speed clinical trials, and comply with federal regulations. “It is the world’s largest clinical registry,” William Rich III, MD, past president of the American Academy of Ophthalmology, said of the IRIS Registry, which exceeded expectations when the number of patients included hit 61 million in November 2015.4
Although glaucomatologists continue to make progress in detecting the disease, treating affected patients, and preventing blindness, large gaps remain in their understanding of glaucoma, its management, how to prevent vision loss, and how to deliver high-quality and cost-effective care. These gaps range from understanding the disease’s impact on patients and their families to determining the effectiveness of glaucoma screening in the community.
Watch it Now
One example of how big data and medical analytics can improve understanding of disease is a longitudinal observational study of 72,587 Medicare beneficiaries in the general community that used the Medicare Current Beneficiary Survey (2004-2009). Researchers found that patients disabled by glaucoma incurred health care costs that were $5,000 higher per year than risk-adjusted nonglaucoma patients. The investigators concluded that reducing the prevalence and severity of the disease might result in improvements in associated nonglaucomatous medical conditions (depression and falls) and reduce health care costs (see video).5
When using big data, it is important to understand its limitations such as confirmation bias, the difference between causation and association, and the inability to follow patients from one database to another. In addition, researchers must bear in mind that it is easy to find random relationships when using large sample sizes.
THE PROMISE OF AI
AI algorithms using deep learning are showing great promise in medicine. For example, a deep learning algorithm did as well as board-certified dermatologists at identifying skin cancer.6 The investigators in this study trained the AI with 130,000 images of skin lesions. In another study, an AI algorithm based on deep machine learning had high sensitivity and specificity for detecting referable diabetic retinopathy.7
AI companies are now working on pattern recognition and deep learning to diagnose abnormalities in the optic nerve (see Deep Learning in Glaucoma). Unlike diabetic retinopathy, glaucoma diagnosis is not only based on optic disc photographs and optical coherence tomography scans. In addition, clinicians consider IOP measurements, glaucomatous optic nerve changes, corneal thickness, and visual field changes. Longitudinal changes are also key to the diagnosis and management of glaucoma. The ideal AI algorithm for glaucoma and other ocular diseases will provide recommendations on diagnosis and management that are highly personalized to the individual’s risk of disease progression and blindness. The approach will be based on risk calculators using big data, data analysis, and deep learning to result in what is called precision medicine.
Deep Learning in Glaucoma
By Sameer Trikha, MBA, FRCOphth; Nicolas Jaccard, PhD; and Jay Lakhani, MEng



Machine learning enables computers to learn without being explicitly programmed to do so. In most cases, machine learning is carried out in a supervised setting, whereby the computer is shown examples belonging to different categories, such as cats and dogs, and tasked with learning how to distinguish between them. This can be done using artificial neural networks, the function of which is inspired by the way the human brain processes information. The most promising method is termed deep learning, which consists of stacking a large number of layers made up of those artificial neurons.1 As the information traverses these different layers, it is increasingly abstracted through a large number of transformations and combinations, giving rise to a new representation of the information that allows the system to make accurate predictions.
Deep learning enables a better-than-human level of performance across a wide range of tasks, including image2 and voice recognition.3 For the first time, these methods can provide computers with a useful and performant artificial intelligence (AI), which will drive a revolution across all sectors, including ophthalmic care.
A CRITICAL JUNCTURE
Growing and aging populations, coupled with newer treatments, are leading to an exponential increase in the demand for health care, with a spiraling of costs. A simultaneous global shortage of trained ophthalmologists is resulting in a huge deficit in high-quality care provision. An estimated 80 million people will have glaucoma by 2020, yet a large proportion of them will not know that they have the disease.4 The early identification of glaucoma is therefore paramount to preventing vision loss, and it requires accurate assessment of the optic disc using retinal imaging in the screening context. In many countries, screening simply does not exist, however, and in others, nonspecialists may screen patients, with false positive rates ranging from 40% to 60%.5
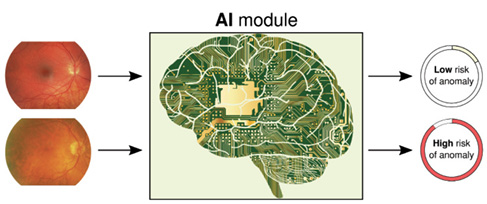
Figure. Teaching computers to spot anomalies in fundus images. An AI module was trained on many thousands of examples labeled by human experts, and it can reliably, consistently, and rapidly predict the degree of anomaly in previously unseen images.
ENTER AI
Based in London, Visulytix (www.visulytix.com) is developing AI clinical decision support systems to provide health care professionals with above-human levels of diagnostic accuracy, delivered in a fraction of a second, to benefit clinical workflow. The goal is to improve the quality of care at the outset, while helping to reduce costs and increase efficiency (Figure).
The company’s Pegasus retinal AI platform autonomously screens for glaucoma through optic disc assessment in addition to classifying the stage of diabetic retinopathy. The most recent results (Visulytix, unpublished data, 2017) indicate that Pegasus can autonomously detect optic nerve abnormalities consistent with potential glaucoma with a sensitivity surpassing that of experts (shown previously to be 80%5). The platform is now being tested at academic centers and by health care providers around the world.
CONCLUSION
AI is likely to become commonplace in ophthalmic care within the next few years, but the technology is unlikely ever to replace eye care specialists and health care providers. Rather, it will augment their decision making to reduce clinical errors and variability. This will ultimately improve the quality of ophthalmic care for all patients and reduce the burden of vision loss for millions.
1. LeCun Y, Bengio Y, Hinton G. Deep learning. Nature. 2015;521(7553):436-444.
2. Wu R, Yan S, Shan Y, et al. Deep image: scaling up image recognition. arXiv preprint. arXiv:1501.02876 7.8 (2015).
3. Xiong W, Droppo J, Huang X, et al. Achieving human parity in conversational speech recognition. arXiv preprint. arXiv:1610.05256 (2016).
4. Tham YC, Li X, Wong TY, et al, Global prevalence of glaucoma and projections of glaucoma burden through 2040: a systematic review and meta-analysis. Ophthalmology. 2014;121(11):2081-2090.
5. Harper R, Reeves B. The sensitivity and specificity of direct ophthalmoscopic optic disc assessment in screening for glaucoma: a multivariate analysis. Graefes Arch Clin Exp Ophthalmol. 2000;238(12):949-955.
Nicolas Jaccard, PhD
• chief technology officer, Visulytix, London
Jay Lakhani, MEng
• chief executive officer, Visulytix, London
Sameer Trikha, MBA, FRCOphth
• chief medical officer, Visulytix, London
• consultant ophthalmic surgeon, King’s College NHS Foundation Trust, London
• media@visulytix.com; Twitter @visulytix
1. Siegler MG. Eric Schmidt: every 2 days we create as much information as we did up to 2003. TechCrunch. https://techcrunch.com/2010/08/04/schmidt-data/. Posted August 4, 2010. Accessed July 10, 2017.
2. Coleman AL. How big data informs us about cataract surgery: the LXXII Edward Jackson Memorial Lecture. Am J Ophthalmol. 2015;160(6):1091-1103.e3.
3. Olsen L, Aisner D, McGinnis JM. The Learning Healthcare System: Workshop Summary. Washington, DC: National Academies Press; 2007.
4. Rich WL III. Ophthalmology, IRIS Registry, and meaningful use. http://nhpf.org/uploads/handouts/rich-slides_12-04-15.pdf. December 4, 2015. Accessed July 10, 2017.
5. Prager AJ, Liebmann JM, Cioffi GA, Blumberg DM. Self-reported function, health resource use, and total health care costs among Medicare beneficiaries with glaucoma. JAMA Ophthalmol. 2016;134(4):357-365.
6. Esteva A, Kuprel B, Novoa RA, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature. 2017;542(7639):115-118.
7. Gulshan V, Peng L, Coram M, et al. Development and validation of a deep learning algorithm for detection of diabetic retinopathy in retinal fundus photographs. JAMA. 2016;316(22):2402-2410.
Lama A. Al-Aswad, MD, MPH
• associate professor of ophthalmology; director of the Tele-ophthalmology Initiative; glaucoma fellowship director; and chair of quality assurance at The Edward S. Harkness Eye Institute, Columbia University Medical Center, New York
• (212) 305-6709; laa2003@cumc.columbia.edu
• financial interest: none acknowledged

