CASE PRESENTATION
A 63-year-old man with pigment dispersion glaucoma (PDG), mild in the right eye and moderate in the left eye, was referred for a consultation because of bilateral disease progression that occurred between 2018 and 2020.
On examination, unmedicated IOP was 19 mm Hg OU. The optic nerve in the right eye looked relatively healthy, whereas superior thinning was clearly evident in the left eye (Figure 1). Visual field progression was confirmed in the left eye (Figure 2), and progressive damage to the retinal nerve fiber layer of each eye was observed, although the findings were more subtle in the right eye (Figure 3). A laser peripheral iridotomy (LPI) had been performed on the right eye in 2010 and was found to be patent. Visual fields from 2010 were available (Figure 4). Anterior segment imaging confirmed the presence of posterior bowing of the iris in the left eye but not the right eye (Figure 5).

Figure 2. Visual field testing in 2018 (A) and 2020 (B) clearly demonstrates progression in the left eye.

Figure 3. OCT scans from 2018 show possible retinal nerve fiber layer progression in the right eye and definite progression in the left eye.
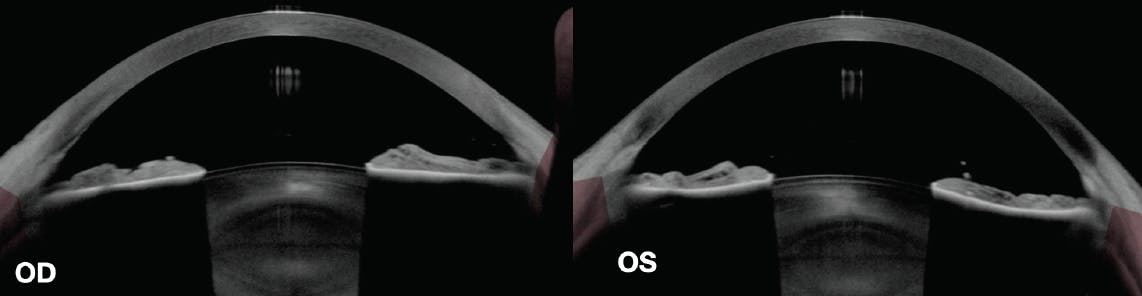
Figure 5. Anterior segment OCT (AS-OCT) imaging of the right eye (at left) shows a flat iris profile after LPI. AS-OCT imaging of the left eye (at right) shows posterior bowing of the iris in the absence of an LPI.
Selective laser trabeculoplasty (SLT) was performed on each eye. IOP subsequently decreased to 16 mm Hg OD but remained at 19 mm Hg OS. Measurements with an iCare Home tonometer (iCare USA) after SLT demonstrated bilateral IOP fluctuation, with mean IOPs of 18.5 mm Hg OD and 21.5 mm Hg OS. Peak IOP readings were 27 mm Hg OD and 29 mm Hg OS, and IOP was generally higher in the left eye than in the right eye (Figure 6).

Figure 6. A report from the iCare Home tonometer shows IOP fluctuations and a higher IOP in the left eye compared to the right.
The patient asks why, given that SLT is often successful in eyes with PDG, the response in his left eye was poorer. How would you respond? Would you repeat SLT, initiate topical medical therapy, or consider a surgical procedure? Would you consider performing an LPI in the left eye because of the better course in the right eye?
—Case prepared by Devesh K. Varma, MD, FRCSC
SHAN C. LIN, MD
There is significant glaucomatous visual field progression in the left eye. Field loss is not evident in the right eye, and optic nerve deterioration is mild on OCT. This asymmetry in disease severity and progression suggests that greater diurnal IOP fluctuation is a cause of the left eye’s worsening status. It is possible that an LPI in the right eye prevented the IOP fluctuation and thus helped preserve the optic nerve. Research suggests that LPI may be effective in preventing future IOP elevation and glaucoma in the early stages of pigment dispersion.1 The benefit of LPI in eyes with existing PDG is controversial. The procedure may be particularly useful if there is active pigment release and/or if showers of pigment occur during exercise.
With regard to SLT, the procedure’s effect on PDG eyes is more variable than on eyes with primary open-angle glaucoma; some reports have shown a paradoxical increase in IOP after SLT on PDG eyes.2 If trabecular meshwork pigmentation is heavy, using lower energy settings and spreading the treatment over different sessions (90º–180º per session) is recommended. It can be explained to the patient that the more advanced disease stage is the likely cause of the left eye’s poor response and, more specifically, that there is greater damage to the outflow system in that eye.
I would recommend medical treatment as the initial approach. The target IOP range would be in the low teens, particularly in the left eye, which has experienced rapid disease progression. In the long term, the goal may be in the range of 10 to 12 mm Hg for the left eye, given the patient’s relatively young age. Filtering surgery may be necessary to achieve this target, in which case care must be exercised to avoid hypotony, which is a common postoperative complication in myopic eyes.
JAMES LIU, MD, AND KELLY W. MUIR, MD, MHSC
The severity of PDG can wax or wane depending on the amount of pigment shed and the duration of the condition. The asymmetry observed in this patient may be a result of the LPI’s relieving the reverse iris bowing and reducing the amount of pigment shed in the right eye. Trabecular meshwork outflow may therefore be more robust in the right eye. Alternatively, this patient may have asymmetric disease, and the LPI is incidental to the asymmetry.
The IOP fluctuation detected at home with tonometry suggests compromised outflow facility in both eyes. This can be further assessed by a water-drinking test3 or tonography.4 The outflow resistance may be at the level of the trabecular meshwork, but chronic trabecular meshwork dysfunction can also cause atrophy of the distal collector channels. In this situation, the efficacy of SLT usually decreases.
We would focus on medical therapy to decrease aqueous production and increase uveoscleral outflow. In our experience, the efficacy of LPI in PDG eyes is underwhelming, consistent with most reports in the literature.1,5-7 We believe, however, that the clinical course of one eye should inform treatment decisions for the fellow eye, and the right eye has experienced less disease progression after LPI. We would therefore consider LPI in this particular case and would prescribe oral acetazolamide to blunt the anticipated IOP spike. We would counsel the patient that LPI may have a minimal effect because of distal collector channel atrophy. If topical measures prove inadequate, we would consider surgical options such as goniotomy and canaloplasty or, if further glaucomatous progression occurs, filtration surgery.
WHAT I DID: DEVESH K. VARMA, MD, FRCSC
Although LPI in PDG eyes helps anatomically to address posterior bowing of the iris, the procedure can cause IOP spikes, and it may not prevent disease progression in the long term. My usual practice, therefore, is not to perform an LPI for pigment dispersion.
In this case, however, the relative stability of the right eye after LPI compared to the left eye prompted me to perform an LPI in the left eye. Postoperatively, the posterior bowing of the iris resolved (Figure 7), but the IOP remained at 20 mm Hg. The patient is scheduled to undergo SLT followed by repeat home tonometry to determine if IOP fluctuations decrease.
1. Gandolfi SA, Ungaro N, Tardini MG, Ghirardini S, Carta A, Mora P. A 10-year follow-up to determine the effect of YAG laser iridotomy on the natural history of pigment dispersion syndrome: a randomized clinical trial. JAMA Ophthalmol. 2014;132(12):1433-1438.
2. Harasymowycz PJ, Papamatheakis DG, Latina M, De Leon M, Lesk MR, Damji KF. Selective laser trabeculoplasty (SLT) complicated by intraocular pressure elevation in eyes with heavily pigmented trabecular meshworks. Am J Ophthalmol. 2005;139(6):1110-1113.
3. Leydhecker W. The water-drinking test. Br J Ophthalmol. 1950;34(8):457-479.
4. Grant WM. Tonographic method for measuring the facility and rate of outflow in human eyes. Arch Ophthalmol. 1950;44(2):204-214.
5. Wang JC, Liebmann JM, Ritch R. Long-term outcome of argon laser iridotomy in pigment dispersion. Invest Ophthalmol Vis Sci. 2001;42(suppl):s560.
6. Scott A, Kotecha A, Bunce C, et al. YAG laser peripheral iridotomy for the prevention of pigment dispersion glaucoma: A prospective, randomized, controlled trial. Ophthalmology. 2011;118(3):468-473.
7. Michelessi M, Lindsley K. Peripheral iridotomy for pigmentary glaucoma. Cochrane Database Syst Rev. 2016;2(2):CD005655.









