CASE PRESENTATION
A 39-year-old woman with high myopia is referred by her ophthalmologist for an evaluation after an optic disc hemorrhage occurs in the patient’s left eye. She has advanced normal-tension glaucoma (NTG) in both eyes. Her maximum recorded IOP is 16 mm Hg OU. She has a history of migraine and a family history (her mother) of glaucoma. Three years ago, the patient was diagnosed with sleep apnea. She has been wearing an oral appliance since then, and a follow-up sleep study found the device to be 89% effective. Glaucomatous progression has not been documented since the patient started wearing the oral appliance.
The patient’s drug regimen includes latanoprost in each eye. On examination, the IOP is 16 mm Hg OU. A lack of response to the drug is suspected, and therapy is discontinued. The patient is instructed to perform home tonometry using the iCare Home (Icare USA). In-office testing finds an IOP of 16 mm Hg, but home tonometry reveals fluctuations, with IOP readings ranging from 10 to 20 mm Hg (Figure 1)

Figure 1. Home tonometry demonstrates IOP fluctuations between 10 and 20 mm Hg over the course of 5 days.
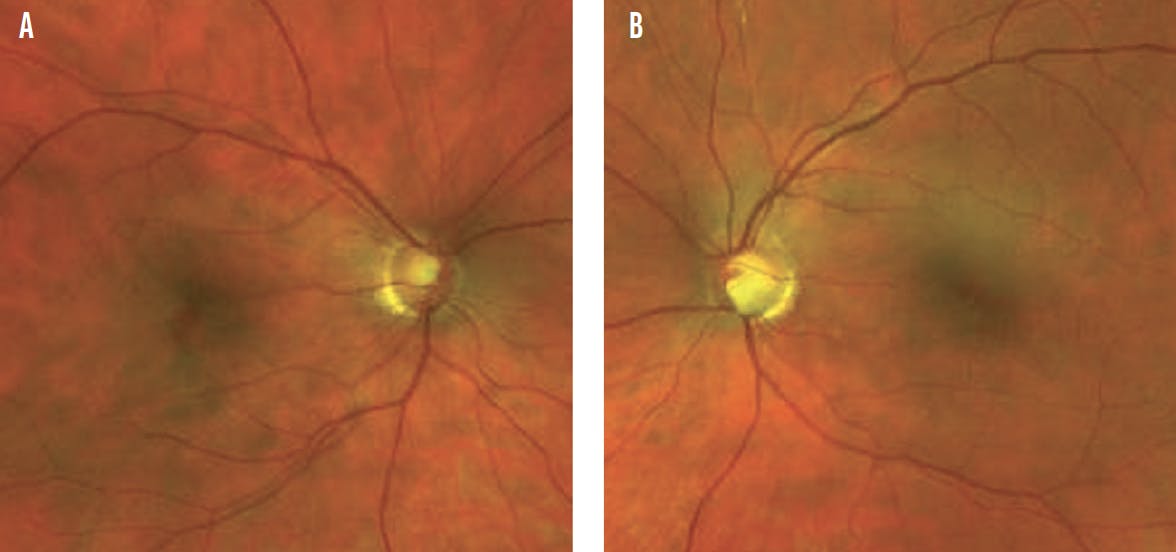
Figure 2. Optic nerve imaging shows superior and inferior rim thinning in the right eye (A) and marked inferior rim thinning in the left eye (B).
The optic nerve of the right eye exhibits superior and inferior thinning, and the optic nerve of the left eye exhibits marked inferior thinning. The optic disc hemorrhage observed by the referring ophthalmologist has resolved (Figure 2). OCT with ganglion cell analysis shows global thinning of the retinal nerve fiber layer (RNFL) that is consistent with high myopia. It also finds focal superior and inferior RNFL thinning in the right eye and inferior RNFL thinning in the left eye consistent with an examination of the optic nerves (Figure 3). Visual field testing demonstrates advanced loss in each eye that splits fixation only in the left eye (Figure 4).

Figure 3. OCT demonstrates global RNFL loss in addition to focal superior and inferior thinning in the right eye and marked inferior thinning in the left eye (A). Ganglion cell analysis shows superior and inferior loss in the right eye and marked inferior loss in the left eye (B).

Figure 4. Visual field testing shows advanced loss in each eye that splits fixation only in the left eye.
Selective laser trabeculoplasty (SLT) is performed bilaterally. Initially, IOP decreases to 14 mm Hg OD and 13 mm Hg OS. Six months after treatment, IOP is 18 mm Hg OU. Visual field testing remains stable.
Given the advanced field and nerve damage, the patient’s age, and her family history, would you offer her additional treatment or recommend observation? If you pursue additional treatment, would you favor topical glaucoma therapy or surgery? If the latter, which procedure? If you would choose observation instead, given the possibility that her optic neuropathy was caused by previous sleep apnea, which is now well controlled, would you proceed to treatment only in the event of definitive disease progression?
—Case prepared by Devesh K. Varma, MD, FRCSC
AHMAD A. AREF, MD, MBA
Given the patient’s young age, high-risk characteristics, and degree of baseline damage, incremental therapy is indicated to reduce the risk of future glaucomatous progression. One risk factor is the IOP fluctuation demonstrated by home tonometry and possibly associated with her sleep apnea. The goal is to lower her peak IOP, which she is likely experiencing overnight.1,2 Nonsurgical therapies that have been shown to lower peak nocturnal IOP include prostaglandin analogues (PGAs), carbonic anhydrase inhibitors, and laser trabeculoplasty.3-5
Therapy with a PGA would be restarted to treat nocturnal peak IOP. I would not base my decision to discontinue this powerful agent on diurnal IOP data. Given the baseline degree of functional deficit and my low tolerance for future loss in this young patient, I would also prescribe a topical carbonic anhydrase inhibitor.
The patient would be monitored closely. She would be scheduled for three to four automated visual field studies annually and office visits every 3 months that include an examination of the optic disc to detect a disc hemorrhage should it recur. If definitive disease progression is noted with formal progression software such as Guided Progression Analysis (Carl Zeiss Meditec) or a new disc hemorrhage is detected, I would recommend incisional glaucoma surgery. My preference would be to start with an angle-based gonioscopy-assisted transluminal trabeculotomy combined with canaloplasty and to consider the placement of a nonvalved aqueous drainage device in the future.
JAMES T. MURPHY, MD
The clinical scenario presented—young patient, low IOP, advanced field loss, cardiovascular risk factors—is one of the more challenging scenarios that a glaucoma specialist encounters. I would emphasize to the patient the importance of lifelong monitoring, but she has many options and, more importantly, has experienced no glaucomatous progression in 3 years.
Latanoprost proved subtherapeutic, but other traditional topical medications as well as netarsudil-containing and latanoprostene bunod drops are alternatives. A lifetime of instilling topical drops is not ideal, but blindness is worse. Considering the current pace of innovation, drops for now may be a more accurate description of her situation than drops for life.
The effect of SLT lasted 6 months, so repeat treatment is a possibility. An IOP of 18 mm Hg is not appropriate for the patient in the long term. Considering the lack of glaucomatous progression, however, since continuous positive airway pressure therapy was initiated, I would monitor her closely. An optic nerve examination would be performed at each visit, and 10-2 visual field testing would be implemented. I would also ask the patient to continue using home tonometry.
Glaucoma specialists strive to stay ahead of disease progression and treat patients proactively. I would nevertheless approach treatment conservatively—at least initially. I would try other drop combinations and repeat SLT in an effort to minimize IOP fluctuation. The patient likely has decades more to live, so I want to be judicious about using conjunctival real estate.
RACHEL G. SIMPSON, MD
A key point in the clinical presentation is that glaucomatous progression has not been documented with OCT or visual field testing for an extended interval following the disc hemorrhage. A conversation with the patient is critical to determining the next steps. If she is reluctant to start treatment and amenable to close observation with regular testing, this may be a reasonable approach. Given her age, family history, field loss, and risk to fixation, I would probably counsel her against an observation-only approach unless she is willing to return for nearly monthly surveillance.
I would recommend a trial of an alternative topical therapy. A Rho kinase inhibitor could be a good choice. Its unique ability to achieve IOP levels lower than episcleral venous pressure is particularly appealing in patients with NTG. It would also be reasonable to try a different PGA because the patient may have a better response.6-8 Whatever topical treatment is tried next, I would ask the patient to use home tonometry to determine the extent of IOP fluctuations and assess the effect of treatment.
I generally reserve invasive incisional glaucoma surgery for patients whose diagnostic imaging documents disease progression. Unfortunately, I find trabeculectomy to be the most successful form of incisional surgery for achieving an IOP low enough to halt the progression of NTG.
WHAT I DID: DEVESH K. VARMA, MD, FRCSC
The patient is young and has significant glaucomatous optic neuropathy. Although she was receiving treatment for sleep apnea, the presence of a new optic disc hemorrhage was worrisome. SLT had a positive but short-lived effect, and I felt that an IOP lower than 18 mm Hg was required. Therapy with preservative-free latanoprost was therefore initiated. The patient tolerated therapy well, and the IOP decreased to 11 mm Hg OD and 12 mm Hg OS. I recommended that she continue latanoprost therapy and return for observation every 3 to 4 months.
1. De Moraes CG, Juthani VJ, Liebmann JM, et al. Risk factors for visual field progression in treated glaucoma. Arch Ophthalmol. 2011;129(5):562-568.
2. Kiekens S, De Groot V, Coeckelbergh T, et al. Continuous positive airway pressure therapy is associated with an increase in intraocular pressure in obstructive sleep apnea. Invest Ophthalmol Vis Sci. 2008;49(3):934-940.
3. Lee AC, Mosaed S, Weinreb RN, Kripke DF, Liu JHK. Effect of laser trabeculoplasty on nocturnal intraocular pressure in medically treated glaucoma patients. Ophthalmology. 2007;114(4):666-670.
4. Liu JHK, Kripke DF, Weinreb RN. Comparison of the nocturnal effects of once-daily timolol and latanoprost on intraocular pressure. Am J Ophthalmol. 2004;138(3):389-395.
5. Liu JHK, Medeiros FA, Slight JR, Weinreb RN. Comparing diurnal and nocturnal effects of brinzolamide and timolol on intraocular pressure in patients receiving latanoprost monotherapy. Ophthalmology. 2009;116(3):449-454.
6. Ren R, Li G, Le TD, Kopczynski C, Stamer WD, Gong H. Netarsudil increases outflow facility in human eyes through multiple mechanisms. Invest Ophthalmol Vis Sci. 2016;57(14):6197-6209.
7. Kazemi A, McLaren JW, Kopczynski CC, Heah TG, Novack GD, Sit AJ. The effects of netarsudil ophthalmic solution on aqueous humor dynamics in a randomized study in humans. J Ocul Pharmacol Ther. 2018;34(5):380-386.
8. Sit AJ, Gupta D, Kazemi A, et al. Improvement of trabecular outflow facility by netarsudil ophthalmic solution in patients with primary open angle glaucoma or ocular hypertension. Poster presented at: ARVO Annual Meeting; May 1, 2019; Vancouver, Canada.






