CASE PRESENTATION
A 78-year-old man presents for an evaluation. The patient has a long history of severe open-angle glaucoma. He underwent a trabeculectomy on each eye 15 to 20 years ago, and the IOP in the right eye has been well controlled ever since. He received a glaucoma drainage device (Ahmed Glaucoma Valve, New World Medical) in the left eye in 2012 to address uncontrolled IOP. The patient later developed corneal edema that required a Descemet stripping endothelial keratoplasty (DSEK) and revision of the glaucoma drainage device, with placement of the tube in the sulcus of the same eye. In 2018, he developed macular edema in the left eye that necessitated a cessation of prostaglandin therapy.
The patient recently presented to the cornea clinic with decreased vision (20/200 BCVA) and an IOP of 25 mm Hg OS. He was found to have corneal edema but no pain or bullae in the eye. Therapy with netarsudil ophthalmic solution 0.02% (Rhopressa, Aerie Pharmaceuticals) was initiated in the left eye, and the patient was asked to follow up with the glaucoma clinic. His current drug regimen, in addition to the netarsudil, consists of dorzolamide, prednisolone acetate, and ketorolac in the left eye and a fixed combination of timolol and brimonidine in both eyes. The patient states that his vision has improved but not to his previous baseline. He adds that he is experiencing superior corneal pain and tearing in the left eye, symptoms that developed after his recent visit.
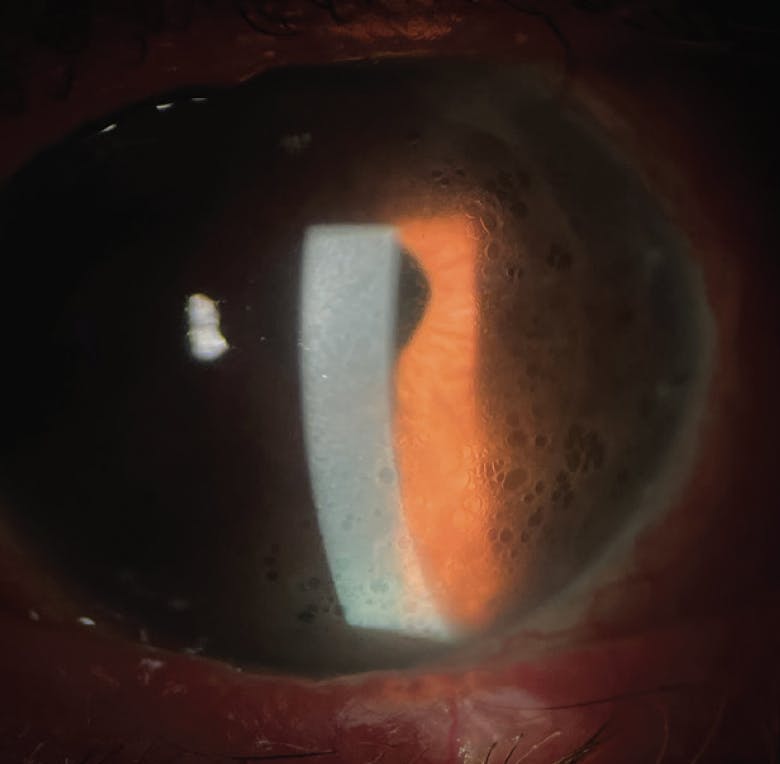
Figure 1. Slit-lamp photography shows mild corneal edema and peripheral honeycomb bullae over a DSEK graft.
On examination, the patient’s BCVA is 20/50 OD and 20/80 OS, and his IOP is 12 mm Hg OD and 19 mm Hg OS. Central corneal thickness is 549 μm OD and 591 μm OS. A slit-lamp examination reveals a well-formed bleb on the right eye and a scarred bleb and a well-covered Ahmed tube in the sulcus of the left eye. The DSEK graft is attached, and central edema and peripheral honeycomb bullae are evident in the left eye (Figure 1). Both eyes are pseudophakic. A fundus examination shows severe cupping of the optic nerve in each eye (Figure 2). OCT imaging shows thinning of the retinal nerve fiber layer in each eye (Figure 3). Humphrey visual field testing (Carl Zeiss Meditec) demonstrates constrictive visual field loss that is greater in the right eye (Figure 4).

Figure 4. Visual field testing demonstrates constriction in the right eye (A) and global depression on total deviation and an inferior arcuate scotoma on pattern deviation in the left eye (B).
The patient desires improved vision and comfort, and he requires a lower IOP. What do you think is the likely cause of his unique pattern of corneal edema? How would you manage this patient?
—Case prepared by Jacob Brubaker, MD
GEORGES M. DURR, MD, FRCSC

Unfortunately, there is no easy solution for this patient. The corneal findings are typical of a rare reaction to netarsudil.1 Several groups have described the reaction, which usually resolves after therapy with the drug is halted. The eye in question has a history of multiple surgeries and is receiving maximal tolerated glaucoma therapy, including dorzolamide and a fixed combination of timolol and brimonidine. The glaucoma is advanced; a significant inferior visual field defect is evident using a 10-2 testing strategy. The target IOP is in the low teens. The IOP is currently 19 mm Hg with netarsudil and will likely rise after treatment with the drug is halted.
A steroid response may be a factor in the patient’s current IOP. To better control the IOP, steroid treatment may be stopped or tapered after a discussion with the corneal surgeon. Therapy with a prostaglandin analogue can also be retried and the patient monitored for signs of macular edema. If these options are not effective, further surgical intervention is likely required. Given the presence of a corneal graft and the previous failure of a glaucoma drainage device, I recommend micropulse or slow-burn cyclophotocoagulation. Compared to a filtering procedure, this intervention poses less risk to the corneal endothelium, but it may exacerbate the macular edema. An alternative is to place a Xen Gel Stent (Allergan) or Preserflo MicroShunt (Santen) in the superior nasal quadrant, but either procedure would increase the risk of corneal graft failure.
MICHAEL D. GREENWOOD, MD

The peripheral bullae and honeycomb pattern were likely caused by netarsudil therapy. The DSEK graft appears to be healthy and not a major contributor to the edema, but this should be evaluated. Therapy with multiple medications is contributing to the patient’s ocular surface irritation and decreased visual acuity. A reduction in the number of agents, if possible, is advisable.
If the macular edema has resolved, I would discontinue treatment with ketorolac and prednisolone acetate but would initiate therapy with loteprednol etabonate ophthalmic gel 0.38% (Lotemax SM, Bausch + Lomb) to protect the DSEK graft. Treatment with the gel should improve the health of the ocular surface and may reduce a steroid response, if present, to the prednisolone acetate.
Next, I would offer the patient a goniotomy with a Kahook Dual Blade (New World Medical) to improve IOP control and perhaps reduce his dependence on IOP-lowering medications. If he still requires treatment with multiple IOP-lowering medications after surgery, I would consider contacting a specialty pharmacy such as Imprimis Pharmaceuticals about combining multiple preservative-free medications in one bottle. This could improve his adherence to prescribed medical therapy and protect the ocular surface.
WHAT I DID: JACOB BRUBAKER, MD

The patient had two problems on presentation: elevated IOP that was poorly controlled despite maximal medical therapy and corneal edema. Each problem made the other worse. The patient had preexisting corneal edema due to corneal graft failure, but he had more recently developed what is termed corneal honeycombing or reticular corneal edema owing to treatment with netarsudil. More common side effects of this drug include conjunctival hyperemia, subconjunctival hemorrhage, and corneal verticillata. Starting 2 years ago, case reports of corneal honeycombing associated with netarsudil therapy began to appear in the published literature.1-6 The etiology of this side effect is unknown, but it seems to occur more frequently in patients who have preexisting corneal edema or uveitis and those with a history of anterior segment surgery. In most of the cases reported to date, the cessation of netarsudil therapy was curative.
The patient had experienced a modest IOP response to netarsudil therapy, but the target IOP had not been achieved. Moreover, the treatment had caused painful peripheral bullae and worsened his corneal edema. He required a lower IOP to prevent glaucomatous progression and to allow a repeat endothelial keratoplasty to be performed.
Netarsudil was immediately discontinued, and surgical options were discussed with the patient. The alternatives included the placement of an additional tube shunt and micropulse transscleral laser therapy (MP-TLT). Because the patient had a history of multiple conjunctiva-based surgical procedures and his cystoid macular edema had been well controlled since the cessation of prostaglandin therapy, we elected to proceed with minimal MP-TLT. The total duration of treatment was 200 seconds with a sweep speed of 10 seconds per quadrant, a power of 2,500 mW, and a duty cycle of 33.3%.
One month after MP-TLT, the IOP was 12 mm Hg OS on a therapeutic regimen of timolol, brimonidine, and dorzolamide. The patient’s visual acuity was counting fingers due to diffuse corneal edema. He subsequently underwent Descemet membrane endothelial keratoplasty.
Four months after MP-TLT and 2 months after Descemet membrane endothelial keratoplasty, his visual acuity was 20/50 OS, and the IOP in that eye was in the midteens. The cornea was clear, and there was no evidence of cystoid macular edema.
This case exemplifies how a stepwise approach can achieve a desirable result.
1. Moumneh K, Sheybani A, Fellman RL, Godfrey DG, Grover DS. Reticular corneal edema or corneal honeycombing in eyes treated with netarsudil: a case series. J Glaucoma. 2020;29(7):607-610.
2. Ramakrishnan MS, Addis VM, Lehman AY, Sankar PS. Netarsudil-associated epithelial keratopathy. Am J Ophthalmol Case Rep. 2020;19:100800.
3. Wisely CE, Liu KC, Gupta D, Carlson AN, Asrani SG, Kim T. Reticular bullous epithelial edema in corneas treated with netarsudil: a case series. Am J Ophthalmol. 2020;217:20-26.
4. Chen H, McMillin JC, Frankfort BJ, Al-Mohtaseb Z. Reticular epithelial edema: an uncommon side effect of ROCK/NET inhibitor netarsudil. J Glaucoma. 2020;29(11):e124-e126.
5. LoBue SA, Moustafa GA, Vu A, Amin M, Nguyen T, Goyal H. Transient reticular cystic corneal epithelial edema with topical netarsudil: a case series and review. Cornea. 2021;40(8):1048-1054.
6. Chu MJ, Song M, Palmares T, Song A, Song J. Rhopressa-induced corneal edema: a case report. J Med Case Rep. 2021;15:182.







